Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Energy Storage
Crab Shells Help Researchers Make New Battery Materials
Lithium Batteries: The nanostructures of crab shells provide a cheap, renewable template for sulfur and silicon electrode materials
by Leigh Krietsch Boerner
June 20, 2013

Crab shells usually are just a nuisance that you have to crack and dig through to get the delicious meat inside. But one team of materials scientists thinks the shells could help them fabricate materials for long lasting batteries. The team used the nanostructures found in the crustacean shells as templates to make sulfur and silicon electrode materials for lithium-ion batteries (Nano Lett. 2013, DOI: 10.1021/nl401729r).
Most lithium-ion batteries on the market have lithium cobalt oxide cathodes and carbon anodes. If those batteries used sulfur as the cathode or silicon as the anode instead, the batteries’ theoretical energy storage capacity would be 10 times greater, says Yi Cui, a materials scientist at Stanford University.
Unfortunately, electrodes made from those materials have structural and chemical problems that lead to short battery lives. For example, as the battery discharges and recharges, the electrodes expand and contract as the sulfur or silicon reacts with lithium ions in the battery electrolyte. This repeated expansion and contraction cracks the electrodes.
To counter this problem, some researchers, including Cui, have tried to contain the sulfur or silicon inside nanostructures such as carbon nanofibers (Nano Lett. 2011, DOI: 10.1021/nl2027684). These structures have enough room for the electrode materials to expand and contract freely, Cui says. Researchers make these nanofibers by growing them on templates, often made from aluminum oxide.
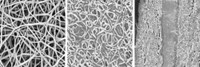
Cui and his team thought crab shells could serve as an inexpensive, environmentally friendly alternative to those templates. Previous studies showed that the shells consist of about 70-nm-wide calcium carbonate nanochannels—just the right size to contain electrode materials. Every year, the food industry throws away about 500,000 tons of crab shells, making the crustacean waste a low-cost, renewable source of these templates.

To test their idea, Cui and his group bought stone crabs from a local Chinese grocery store. After collecting the crabs’ shells, they prepared the templates by first burning off organic matter on the shells and then grinding the calcium carbonate shells into a powder. Next they mixed the powder in a solution of dopamine to coat the calcium carbonate in a dopamine polymer. Heating the powder at 800 °C turned the polymer into a layer of carbon. Finally, using dilute hydrochloric acid, the scientists etched away the calcium carbonate shell. What remained were hollow, 65-nm-wide carbon nanofibers grouped into bunches, Cui says.
The researchers stuffed these nanostructures with sulfur by heating them with the element under argon, or filled them with silicon via chemical vapor deposition. They then made electrochemical devices to test each material’s performance as an electrode.
As a test of the electrodes’ lifetime, the team put them through 200 charge-discharge cycles. After those cycles, 60% of the energy storage capacity remained in the sulfur-containing electrode, and 95% remained in the silicon one. Cell phone battery electrodes retain about 80% of their capacity after 500 cycles, Cui says. However, he points out that it’s difficult to make a full comparison with commercial electrodes because the test devices were not full batteries.
A cheap, renewable template for high-capacity electrode materials is an important advance, says Galen D. Stucky, a materials scientist at the University of California, Santa Barbara. But he says that the researchers’ materials don’t retain enough of their high energy-capacity over time to be ready for practical applications, such as use in a hybrid car.
Cui says his group is now working on other biological templates, such as ones made from rice husks, and trying to improve the ability of the electrodes to store charge.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter