Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Molecular Motor Spins In Two Directions
ACS Meeting News: A ruthenium-based complex spins clockwise or counterclockwise, depending on how electrons tunnel through it
by Leigh Krietsch Boerner
September 11, 2013

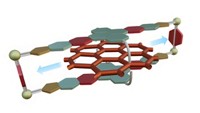
Inside cells, tiny molecular motors spin to synthesize biomolecules or walk to transport cellular cargo. Scientists want to copy these motors to make nanomachines, but so far synthetic motors have spun intermittently and in only one direction, limiting their usefulness. This week, during a session in the Division of Inorganic Chemistry at the American Chemical Society national meeting in Indianapolis, scientists reported a ruthenium-based motor that not only spins consistently in one direction, but also can reverse itself.
The inorganic complex, synthesized by Gwénaël Rapenne, an inorganic chemist at the University Toulouse, in France, and his colleagues, looks like a piano stool. A ruthenium atom sits at the center of the stool, sandwiched between two ligands. On the bottom, a hydrotris(indazolyl)borate ligand serves as a tripod for the motor. Its sulfur groups can anchor the complex to a gold surface. The top ligand is a cyclopentadienyl ring connected through benzene rings to four ferrocenes and one methyl group. This ligand acted like a rotor, spinning on the ruthenium atom.
To make the motors spin, the team cooled a gold surface decorated with the complexes to 5 K, and then used the tip of a scanning tunneling microscope (STM) to direct electrons at either a ferrocene or the methyl group on the rotor. They followed the rotors’ movements using the STM and found that when an electron hit a ferrocene group, the rotor spun counterclockwise. But when the electron struck a methyl group on the rotor, it rotated clockwise (Nature Nanotech. 2013, DOI: 10.1038/nnano.2012.218).
This is the first example of a synthetic molecular motor that can reverse its direction of rotation, Rapenne says. The rotation was also consistent: The rotor spun in the same direction 88% of the time it was hit by an electron. The most successful past molecular rotor spun in one direction only 5% of the time.
The complex can spin in either direction because of the asymmetrical shape of the rotor, Rapenne says. When an electron hits the rotor it jumps from the ligand to the gold surface through a process called electron tunneling. The way the electron tunnels depends on which group on the rotor it hits, due to the difference between the electron orbitals of the ferrocene and methyl groups. This tunneling difference pushes the rotor in opposite directions.
Marilyn R. Mackiewicz, a nanochemist at Portland State University, is impressed by how Rapenne and his colleagues figured out the mechanics behind their motor’s movement. The fundamental aspects of what makes the rotor spin are very important to driving the field forwards, she says.
Rapenne says his group will next try to hook up their motor to a nanosized winch to see if it can do mechanical work. He envisions using these motors to turn winches or move molecular cargo in nanomachines.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter