Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Spectroscopy
Detecting Molecules Through Bone
Brain Imaging: Raman spectroscopy detects chemicals through bone and may eventually provide a noninvasive way to peek inside the brain
by Sarah Webb
November 20, 2013
To understand the brain and its chemical complexities, researchers would like to peer inside the skull and observe neurotransmitters at work. Unfortunately, research methods to measure levels of certain chemicals in the brain require drilling holes in the skull. Some neuroscientists have started to develop ways to use magnetic resonance imaging to monitor specific molecules. Now, as a first step toward another noninvasive imaging tool, researchers have shown that they can use Raman spectroscopy to detect chemical signatures through bone (J. Am. Chem. Soc. 2013, DOI: 10.1021/ja409378f).

With Raman spectroscopy, chemists can look for chemicals of interest sitting inside a range of materials, such as explosives inside plastic bottles. Richard P. Van Duyne’s group at Northwestern University has used the technique to monitor glucose levels through the skin of living rats (Anal. Chem. 2011, DOI: 10.1021/ac202343e). Bhavya Sharma, a postdoc in the group, wondered if the same Raman technique also could work through bone. Such an analytical method could allow scientists to watch as levels of neurotransmitters change in the brain, a feat that currently requires drilling into the skull and inserting probes.
To peer through bone, Sharma and colleagues combined two spectroscopic techniques: surface-enhanced and spatially offset Raman spectroscopy. Both methods involve exciting samples with laser light and then monitoring for specific Raman signals from the sample that are characteristic of a chemical of interest.
In the surface-enhanced variety, gold nanoparticles boost the Raman signal produced by molecules bound to their surfaces. The spatially offset method allows researchers to detect a useful signal from molecules located up to 20 mm within a sample. Researchers can isolate signals from these buried compounds by observing Raman signals at a different spot from where they shine the laser light. The separation ensures that the molecule’s signal isn’t dwarfed by scattered laser light from the sample’s surface.
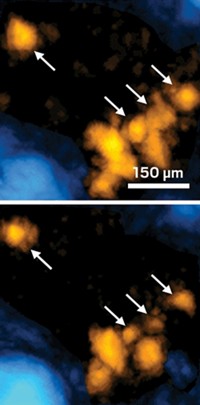
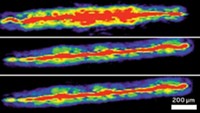
As a test of the combination method, the researchers went to the market and bought a cut of lamb shoulder with a bone 3 to 8 mm thick. The human skull is 3 to 14 mm thick. The team then injected 90 trillion gold nanoparticles into the meat behind the bone. They had decorated the particles with a compound that has a strong Raman signature. When they shined 785-nm laser light on the bone, they could immediately detect the chemical signature of the reporter molecule. Sharma jumped up and down when she saw the results. “Everything I read and everyone we talked to said, ‘No, this shouldn’t work,’” through bone because the material isn’t transparent enough, Sharma says.
Right now the researchers cannot detect where the nanoparticles are located within the tissue, Van Duyne says, only that they are on the other side of the bone. And even with further refinements, the depth of tissue penetration is likely to be limited to areas close to the tissue’s outer surface.
The paper is “a nice piece of work,” says W. Ewen Smith of the University of Strathclyde, in the U.K. But he cautions that this paper is only a first step toward a technique that could be clinically useful. One significant challenge will be to develop nanoparticles that can target molecules of interest but are not toxic at the levels needed for detection, he says.
Van Duyne recognizes that his team is at the beginning of a long process to understand how Raman might work as a brain imaging technique. As a next step, the team plans to attach dopamine to the nanoparticles to see if they can detect the neurotransmitter through bone. “This is just step one,” Van Duyne says, “but it’s a showstopper.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter