Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
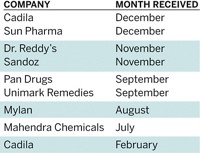
FDA Bans Another Indian Drug Plant
Generics: Quality problems plague Indian producers
by Jean-François Tremblay
March 24, 2014
| A version of this story appeared in
Volume 92, Issue 12
Continuing a crackdown against shoddy foreign drug manufacturing, the U.S. Food & Drug Administration has again banned imports of pharmaceuticals made at a facility in India.
The latest action targets a Sun Pharmaceutical Industries plant in Karkhadi, Gujarat, that produces the antibiotic cephalosporin. Sun is one of India’s largest drug producers.
INDIAN PHARMA
Achievements And Notoriety
◾ Number of warning letters U.S. FDA issued to Indian firms in 12 months through September 2013: 7 (Total letters to non-U.S. firms: 26)
◾ Number of Indian firms on Europe’s noncompliance list: 35
◾ India’s share of FDA applications for generic drugs in 2012: 40%
◾ Number of Indian plants allowed to export drugs to the U.S. in 2011: 120
◾ Biggest U.S. fine against an Indian firm: $500 million (for Ranbaxy, for manufacturing deficiencies)
SOURCES: FDA, Drug Quality Assurance, India Brand Equity Foundation, USA-India Chamber of Commerce
According to Sun, FDA’s action took place after a recent inspection of the facility during which agency officials identified instances of noncompliance with the regulatory standards known as current Good Manufacturing Practices, or cGMP. The Karkhadi facility accounts for only a small portion of Sun’s sales, the firm says.
FDA hasn’t revealed what manufacturing deficiencies its inspectors found, but the problems were likely serious, according to Peter Saxon, president of Saxon International Associates, a New Jersey-based company that advises companies on cGMP compliance. “An import alert is usually issued if the firm falsified documentation or when gross contamination is proved or observed,” he says.
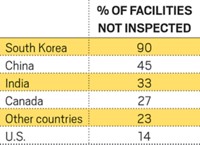
In recent years, India has become one of the largest suppliers of generic drugs to the U.S. But as Indian companies increase their U.S. presence, FDA is finding manufacturing deficiencies at a growing number of their plants.
In 2012, Ranbaxy Laboratories, one of India’s largest drugmakers, agreed to pay $500 million in fines over problems found at several of its facilities. And in December, FDA banned drugs made by Wockhardt, another leading Indian producer.
FDA is beefing up its presence in India. Last month, FDA Commissioner Margaret A. Hamburg visited the country, where she expressed concerns about drug quality (C&EN, Feb. 24, page 6). The agency is now recruiting seven drug investigators to supplement the 12 who are already based in India, an FDA spokeswoman tells C&EN.
“There are many good companies in India but also a significant number that think that cGMP does not apply to them,” Saxon says.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter