Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Alzheimer’s Prion Connection Deepens
Neuroscience: Like prions, hallmark Alzheimer’s molecules fold into various shapes with distinct behaviors
by Lauren K. Wolf
July 7, 2014
| A version of this story appeared in
Volume 92, Issue 27

In the past few years, scientists have demonstrated that molecules involved in Alzheimer’s disease behave like the prion proteins that cause mad cow disease. That is, the Alzheimer’s molecules—amyloid-β peptide and tau protein—misfold in the brain and then coerce other molecules to do the same. The misfolded molecules then clump and damage nerve cells.
Alzheimer’s researchers are now following this trail and finding other similarities between prion proteins and amyloid-β and tau. For example, like prion proteins, amyloid-β and tau form different “strains”—distinct three-dimensional shapes that begin clumping in the brain at distinct times.
This observation might have important implications for Alzheimer’s drug discovery, says Kurt Giles, coleader of two new studies confirming the existence of amyloid-β strains (Proc. Natl. Acad. Sci. USA 2014, DOI: 10.1073/pnas.1408900111 and 10.1073/pnas.1408968111). “It’s possible that some therapeutics might be effective against some strains and not others,” says Giles, a neuroscientist at the University of California, San Francisco.
Along with prion pioneer and Nobel Laureate Stanley B. Prusiner, also at UCSF, Giles and coworkers showed that they can pass certain strains of amyloid-β from one lab mouse to another.
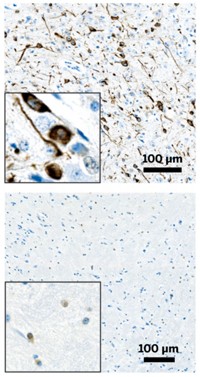
In its first study, the team took brain extracts from two deceased Alzheimer’s patients: One had a so-called Arctic mutation in the gene that produces amyloid peptide, and the other had what is known as a Swedish mutation. When the researchers injected these extracts into the brains of mice, they observed the Arctic extract trigger the characteristic formation of fuzzy-looking amyloid clumps and the Swedish extract generate thin, compact clumps. The Arctic aggregates appeared in the rodents’ brains more quickly than the Swedish aggregates.
When the researchers extracted brain tissue from the mice and injected it into the brains of a second generation of rodents, they observed the same onset times and amyloid pathologies for each type of aggregate, thus indicating the existence of strains.
In a second study, the UCSF team made aggregates of two synthetic amyloid-β fragments, one containing 40 amino acids and the other containing 42. In solution, amyloid-β42 clumps into short fibrils, and amyloid-β40 clumps into both long and short fibrils. When injected into the brains of mice, these two solutions induced the formation of small and large aggregates, respectively.
Because amyloid-β40 and amyloid-β42 are different peptides and because the UCSF team didn’t link them to unique molecular structures, it isn’t clear whether they should be called strains, comments Marc I. Diamond, a neurologist at Washington University in St. Louis. But, he says, the team did demonstrate the strain concept by propagating the Arctic and Swedish aggregates from mouse to mouse. Diamond and his team recently proved the existence of tau protein strains (Neuron 2014, DOI: 10.1016/j.neuron.2014.04.047).
Neil Cashman, an expert in protein misfolding diseases at the University of British Columbia, says, “These papers provide compelling experimental support” for the idea that Alzheimer’s precursor molecules act like prions.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter