Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Microwave Chemistry Remains Hot, Fast, And A Tad Mystical
Differing takes on exactly how this popular heating method enhances reactions is keeping chemists intrigued
by Stephen K. Ritter
January 27, 2014
| A version of this story appeared in
Volume 92, Issue 4

When microwave ovens started appearing in chemistry labs in the late 1980s, it wasn’t just to heat up leftovers for lunch or to make popcorn or a frozen burrito for a late-night snack. Imaginative organic chemists had figured out that there’s an advantage to using microwaves to rapidly and uniformly heat reaction mixtures to accelerate synthetic processes. The word quickly spread to materials scientists making nanoparticles, biochemists synthesizing peptides, and environmental scientists preparing samples for chemical analysis.
Microwave reactors are now standard equipment throughout the pharmaceutical and chemical industries, as well as in many university organic synthesis and teaching labs. Organic chemists are particularly enamored with microwaves because reactions that take hours of refluxing in a flask sitting in an oil bath at atmospheric pressure can instead be completed in minutes, often with improved yields and product purity.
“Process intensification at high temperature and pressure is the essence of microwave chemistry,” says C. Oliver Kappe of the University of Graz, in Austria, one of the leaders in the field. “That is why a microwave reactor can be a powerful tool.”
But there is a “but.” Although the ability to carry out hot, fast reactions using microwaves has attracted dedicated practitioners, there has always been some confusion about how microwave chemistry works. After years of careful experiments, scientists agree that microwaves induce only a thermal effect—that is, microwaves are just an efficient way to heat reactions and no magical radiation effect is at play. However, new results have suggested that microwaves might be inducing subtle thermodynamic effects that aren’t connected to a change in bulk reaction temperature. An acerbic debate erupted last year among some researchers in the field—including Kappe—on how to interpret the new findings and whether they might someday lead to practical chemical processes.
In a nutshell, microwave heating of homogeneous solutions occurs when an oscillating microwave electromagnetic field interacts with polarizable molecules or ions. The energy of microwaves is too low to directly activate or break molecular bonds. Microwaves therefore can’t induce molecules to undergo chemical reactions by direct absorption of energy in the way that higher energy visible and ultraviolet light can in photochemical reactions. Microwave energy must first be converted into heat.
Heating occurs as the polarized species fight to align with the electromagnetic field: They rotate, bump into, and rub against each other, which causes them to warm up directly without having to heat the reaction vessel. This microwave effect differs from conventional heating in which the reaction vessel is heated and the vessel transfers the heat to the reaction mixture indirectly by convection.
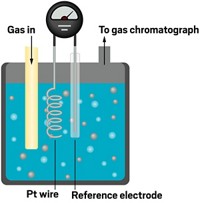
During microwave chemistry’s early years, researchers used ordinary kitchen microwave ovens and open beakers, so they had a hard time monitoring and controlling the temperature and pressure. Inconsistent results led chemists to debate the reasons for the observed reaction rate enhancements and product selectivities and to wonder whether magical nonthermal microwave radiation effects were at play. These effects were postulated to result from a direct stabilizing interaction of microwaves with specific molecules, intermediates, or transition states.
Kappe’s group has helped lead the way over the past decade in assessing the plausibility of nonthermal microwave effects and some proposed specific thermal microwave effects, debunking many claims of extraordinary results. His team has shown through careful reinvestigations that in most cases erroneous temperature measurements were to blame or the experimental conditions biased the results.
But some scientists have observed unexplained specific microwave effects. These effects appear to be a result of the rapid heating altering normal thermodynamic processes, leading to enhanced reactivity or enantioselectivity that is not connected to an increase in the bulk reaction temperature.
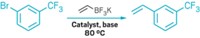
One of these examples was reported in 2012 by Gregory B. Dudley, Albert E. Stiegman, and coworkers at Florida State University. The researchers carried out a Friedel-Crafts benzylation of deuterated p-xylene in an open vessel at constant microwave power using a highly polar benzyl-containing pyridinium salt as the precursor. The xylene, which is nonpolar, doubled as a reactant and a non-microwave-absorbing solvent (Chem. Sci. 2012, DOI: 10.1039/c2sc01003h).

This reaction system was designed to provide a way to see if microwaves might be capable of selectively heating individual solution components. The researchers note that their bulk homogeneous solution in reality appears heterogeneous at the molecular level, where the ionic reactant molecules are transiently encased by solvent.
This situation is analogous to known microwave heterogeneous reactions in which a solid catalyst is selectively heated and the gas reactants are microwave transparent and remain cold. Materials scientists, for example, take advantage of this type of selective heating when making nanoparticles—it often enables smaller particle sizes, narrower size distributions, and a higher degree of crystallization compared with conventional convective heating.
Dudley, Stiegman, and coworkers found that microwave irradiation significantly increased the benzylation reaction rate relative to the same reaction carried out at about the same temperature using standard oil-bath heating. But that result goes counter to the expectation that an increase in reaction rate must be tied to an increase in bulk temperature.
The Florida State researchers acknowledge that their example is an extreme case and that more work is needed to provide a satisfactory explanation for their observations. But they favor the idea that the rapid heating from microwave irradiation could alter the type and frequency of molecular collisions of the polar molecules, enhancing the reaction rate without increasing the bulk temperature.
“This type of system could provide a foundation for the rational design of new microwave-actuated organic reactions for strategic applications in chemical synthesis,” Dudley says.
To understand this idea, one has to dust off their physical chemistry textbook and refer to the Arrhenius equation, k = Ae–Ea/BT, which defines the relationship between temperature and chemical reaction rates (k). It includes activation energy (Ea) and the Boltzmann constant (B), which relates the energy of the system with temperature. These entities are combined in the exponential term, which gives the probability of molecules reacting.
The Arrhenius equation also has a preexponential factor (A), which is a constant unique for different chemical reactions. Like other constants, it can be determined experimentally. The value of this constant depends on how often the reactant molecules collide and on whether they are properly oriented to react when they collide.
Dudley and Stiegman think the selective microwave heating in their system could be creating molecular-level, thermally induced differences in collision frequency to increase the reaction rate in a way that is not reflected in the measured temperature. In other words, the reactants are locally very hot and react faster than expected on the basis of the bulk reaction temperature, which is lower because the nonpolar solvent warms more slowly than the polar reactant molecules. This finding suggests that selective heating affects the magnitude of the preexponential factor, allowing the reaction to more readily overcome the energy barrier of the reaction.
“This is not a controversial concept in heterogeneous reactions,” Stiegman says, “where it’s readily understood that a solid is being selectively heated by microwaves.”
Kappe’s group repeated the Dudley-Stiegman experiments and instead attributed the unexpected results to erroneous temperature measurements. Kappe believes the enhanced conversions in the benzylation reaction “are most likely the result of microwave-assisted solvent superheating under atmospheric conditions, and therefore are the consequence of an increased bulk temperature of the reaction mixture.” Put another way, he is suggesting that solvent hot spots, not microwave-altered collisions, lead to the enhanced reactivity (Angew. Chem. Int. Ed. 2013, DOI: 10.1002/anie.201204103).
The Dudley-Stiegman team called foul after Kappe’s assessment of their work. The two sides subsequently traded verbal jabs in a running dialogue that played out in point-counterpoint fashion during the past year in Angewandte Chemie International Edition (DOI: 10.1002/anie.201301539 and 10.1002/anie.201304368).
Neither party believes that nonthermal microwave effects exist. Where they disagree is on the exact nature of specific thermal microwave effects and whether they could be of practical relevance to chemists.
“Microwave-induced atmospheric superheating under open-vessel conditions is difficult to control and maintain, isn’t applicable to larger volumes for safety reasons, and requires a significant amount of microwave power,” Kappe notes. He doubts selective heating would allow much flexibility or provide unique selectivity in synthetic reactions that could not be achieved by simply running the experiment at a higher bulk reaction temperature.
Dudley acknowledges that may be true. “Specific microwave effects may be rare and difficult to design successfully into experiments at this time,” he says. “But there is no reason to doubt that they are possible and that chemists should explore how they might use them to harness microwave energy strategically.”
Dudley and Stiegman report some progress on identifying practical preparative-scale organic reaction systems that could be selectively actuated by microwaves. “We have nothing yet that I would describe as being of immediate synthetic utility,” Dudley says.
The Florida State researchers are not alone with their difficulties in elucidating microwave heating effects. Tohru Yamada of Keio University, in Japan, and coworkers have reported what they call an “extraordinary microwave effect” occurring for several catalytic enantioselective reactions. In these reactions, the reaction rates using microwaves increase compared with conventional heating, which is not unexpected. But the rates increase without any loss of enantioselectivity, which would seem to violate the Arrhenius equation.
In the Arrhenius equation, an inverse relationship exists between reaction temperature and enantiomeric excess—the higher the reaction temperature or the faster the reaction rate, the lower the enantiomeric excess in a reaction. In the Yamada group’s reactions, the enantioselectivities were undiminished, even when the reactions were accelerated 10 to 1,000 times, he says.
Yamada’s group initially reported this effect in enantioselective catalytic ring-opening reactions of biaryl lactones. To confirm those results, the researchers followed up with a study on the catalytic enantioselective Claisen rearrangement of enolphosphonates (Chem. Commun. 2013, DOI: 10.1039/c3cc44610g).
They carried out the reactions at the same constant temperature using a heating block. When running the reactions with and without microwave irradiation, they obtained about the same yield and enantiomeric excess of the products. Yet the microwave reactions occurred much faster.
“This enhancement was not caused by a simple thermal effect,” Yamada asserts, “but by some direct microwave effect on the substrate molecules.” Yamada believes that in both cases the microwave irradiation is pushing the thermodynamic equilibrium between conformational isomers of the substrate molecules in the direction of the isomer favored by the catalyst. He proposes that this shift reduces the activation energy and increases the reaction rate.
Florida State’s Dudley is intrigued by the high rate enhancements reported. “Yamada’s results are both hard to explain and hard to ignore,” he says.
Stiegman agrees. He sees parallels between Yamada’s results and the selective heating of the ionic pyridinium reagent in his group’s work. “In complex solutions such as the ones Yamada is using, it is difficult to isolate a specific effect,” Stiegman says. “But in my view it is likely due to selective heating of the ionic catalyst, which is consistent with known microwave physics.”
For Kappe’s part, he was initially suspicious of Yamada’s approach, which includes cooling the reactions while using microwaves. “What we know from our experience is that the way the samples were treated in this particular case is very prone to errors in temperature measurement,” Kappe notes.
Yamada says that, given the inverse relationship between enantiomeric excess and reaction temperature, his reactions point to a further idea. The enantiomeric excess in asymmetric microwave reactions can actually be used as a kind of thermometer to confirm the average reaction temperature. He suggests doing so would provide an extra metric to help determine any experimental error in temperature measurement.
After discussing the research and this point with Yamada, Kappe says the results “remain unexplainable.” To be sure, he adds, his group or others would have to repeat the experiments to help determine whether there is a specific microwave effect of any kind involved.
Advertisement
“There have been few reports on microwave-assisted enantioselective reactions, and the complete understanding of microwave heating has never been clarified,” Yamada says. “We believe our observation could open a new paradigm and application of microwave-assisted synthetic chemistry.”
“Selective microwave heating is not being consciously exploited in organic synthesis,” Dudley adds. “Scientists are not looking carefully enough for these subtle effects. But this is an area in which basic research leading to better understanding will move science forward.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter