Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
A Backdoor Route To SF5 Heterocycles
Chemists develop an oxidative sulfur fluorination method as an alternative to starting with fluorinated sulfur precursors
by Stephen K. Ritter
November 24, 2014
| A version of this story appeared in
Volume 92, Issue 47
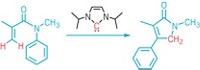
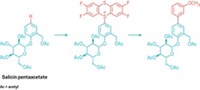
The trifluoromethyl group is an important electronegative substituent that helps control solubility, binding, and metabolism of agricultural chemicals and pharmaceuticals and control properties of electronic materials. But a sulfur pentafluoride group on an aromatic ring is even more electronegative than a CF3 group, making SF5 an especially attractive functional group in agricultural, pharmaceutical, and materials science arenas. Previous approaches to introducing SF5 groups into organic molecules required using SF5-substituted aryl or alkyne reagents or SF5Cl. Oleksandr S. Kanishchev and William R. Dolbier Jr. of the University of Florida now report that excess oxidative fluorination of sulfur in 2,2´-dipyridyl disulfides with a KF/Cl2/acetonitrile system leads in two steps to SF5-substituted pyridines without the need for fluorinated sulfur precursors (Angew. Chem. Int. Ed. 2014, DOI: 10.1002/anie.201409990). Treating the disulfides with the fluorinating system at first leads to SF4Cl groups, the researchers explain. They finish off the fluorination with AgF to obtain the SF5 groups. “This is the first preparatively simple and readily scalable example of the transformation of an existing heterocyclic sulfur functionality to prepare SF5-substituted heterocycles,” the Florida chemists write.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter