Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Physical Chemistry
A Better Measure Of Van der Waals Forces
Study finds that interaction of organic molecules with a gold surface increases with greater electron delocalization
by Jyllian Kemsley
December 8, 2014
| A version of this story appeared in
Volume 92, Issue 49
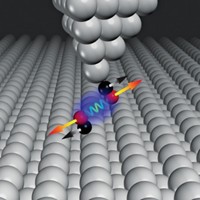
Van der Waals interactions are attractive forces that arise from fluctuations in electron distribution. Such forces are weak and difficult to study experimentally. Nevertheless, they are critical to many areas of chemistry, such as in molecular self-assembly and material adhesion. A team led by Christian Wagner of Germany’s Jülich Research Center has measured for the first time the long-range van der Waals force between molecules and a surface (Nat. Commun. 2014, DOI: 10.1038/ncomms6568). They did the experiments by using an atomic force microscope tip to bind to a single π-conjugated polynaphthalene molecule lying flat on a gold surface, then using the tip to drag the molecule into a vertical position and away from the surface. The interaction between the molecule and the surface was reflected in the oscillation frequency of the AFM tip. Investigating three different polynaphthalenes, Wagner and colleagues found that the van der Waals interactions increased nonlinearly with size such that larger molecules had about 10% greater attraction to the surface than expected from their number of atoms. The researchers think that’s likely because greater electron delocalization makes the molecules easier to polarize. The findings confirm results from computationally expensive modeling and point to a need to incorporate the effects in more routine computations, Wagner says.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter