Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Building A Better Gas Mask
Driven by materials chemistry, today’s masks provide ever-broader respiratory protection
by Mitch Jacoby
December 8, 2014
| A version of this story appeared in
Volume 92, Issue 49

One hundred years ago last month, Allied forces stood their ground in Ypres, preventing the larger and better-equipped German army from taking control of that strategic town in western Belgium. The following spring, Germany attacked again in the Second Battle of Ypres, gassing thousands of French colonial troops with chlorine. That infamous event, which marked the first major use of poison gas as a weapon, introduced the world to the devastating effects of modern chemical warfare. The attack also sent scientists and manufacturers scrambling to develop protective gas masks.
A century later, scientists and engineers continue to improve gas masks and gas mask filters. They are drawing on advances in materials and engineering to design masks that are more effective and functional than ever before. And to ensure that the overall gas mask package provides the highest level of respiratory protection, they are studying new types of filtration media, particularly porous metal-organic framework (MOF) compounds.
Unlike in the early-20th century, when gas masks were designed mainly for limited military use, today’s breathing protection devices, known as air-purifying respirators, are standard equipment in numerous markets around the globe, according to Tom Korb, senior global product manager at Melksham, England-based Avon Protection.
Korb notes that more than 2 million Avon respirators are used in some 65 countries. Not only are these kinds of masks used by military personnel, they’re donned by law enforcement agents protecting themselves from pepper spray during riots. They also protect first responders, including firefighters; hazardous materials specialists; workers who may be exposed to toxic fumes and harmful particulates, for example, in the oil, gas, demolition, and mining industries; and people with other occupations, such as spray painting, that call for respiratory protection from solvent vapors.
Air-purifying respirators have also been distributed to civilians under threat of attack by nerve agents, blister agents, and other types of chemical warfare agents. Even today, the threat of such attacks remains high in some places. For example, a United Nations mission concluded that in August 2013, residents of the Ghouta region of Damascus, Syria, few of whom had access to masks, were gassed with the nerve agent sarin. Reported death tolls ranged from hundreds to more than 1,000. Fear of a similar attack sent Israelis at that time to government distribution centers to collect masks and other protective gear.
The earliest mass-produced gas masks were little more than flannel head and neck coverings with a rectangular peephole. These early World War I masks, developed in response to the Ypres attack, were treated with sodium thiosulfate to neutralize chlorine. Not long thereafter, the crude personal protection devices gave way to more advanced masks that were connected to a canister filled with activated charcoal to filter poison gases.
Modern masks have some similarities with the earliest ones. To this day, for example, activated charcoal, also known as activated carbon, remains the most common filter material.
“We have been using activated carbon to remove toxic gases for about 100 years,” says Gregory W. Peterson, leader of a gas mask filtration research group at the Edgewood Chemical Biological Center (ECBC). Located at the U.S. Army Aberdeen Proving Ground in Maryland, ECBC is the leading U.S. military R&D center for chemical and biological defense.
Activated carbon isn’t a single material. The term refers to a family of carbon-based solid sorbents typically prepared by heat-treating coconut shell, wood, coal, and other carbonaceous substances. Owing to their microporosity and high surface areas, these materials trap a broad range of organic compounds and other substances. Suppliers commonly impregnate activated carbons with metal salts, amines, and other species to enhance the materials’ ability to interact with and capture various types of analytes, especially highly volatile ones. Peterson notes that today’s U.S. military gas mask filters use carbon impregnated with copper, silver, zinc, molybdenum, and triethylenediamine.

Despite a century’s worth of experience and success using activated carbons in gas mask filters, these materials cannot protect soldiers and others from all toxic gases under all conceivable conditions. Identifying deadly combinations of poisons and conditions that thwart today’s gas masks in a public forum, such as this magazine, could lead to dire consequences. No responsible person with expertise in gas mask filtration would broadcast that kind of specific information.
The way experts such as Peterson describe activated carbon’s shortcomings is by noting, first of all, that the materials trap gases and impregnants via relatively weak physical adsorption. In addition, to protect soldiers and others against a broad spectrum of toxic gases—acids and bases, for example, or gases that react in the atmosphere and form acidic and basic species—carbon must be treated with a number of chemically distinct compounds. Over time, these species can interact in the solid’s pores and reduce its effectiveness as a filter.
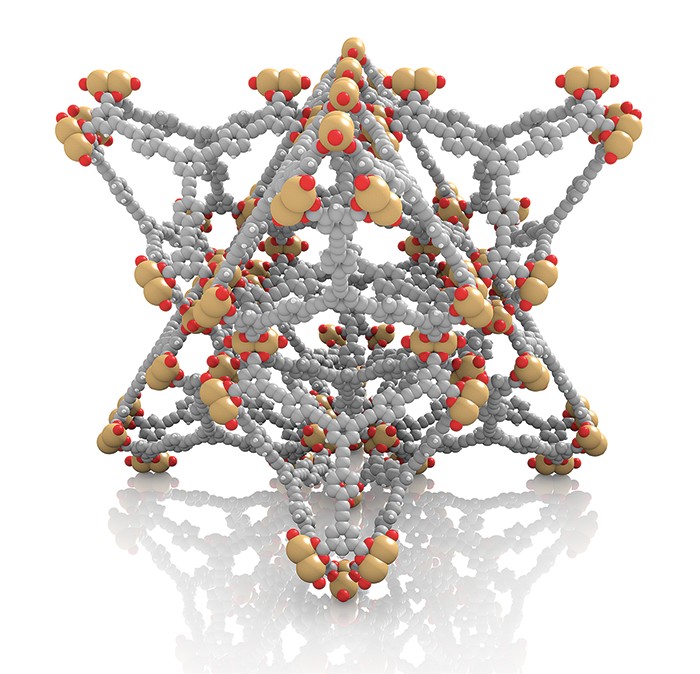
One strategy for improving respiratory protection is to use filter materials that bind analytes tightly, via covalent bonds. That’s one of the reasons MOFs look promising for gas masks. With more than 6,000 examples, they form a large family of porous crystalline materials composed of metal ions or metal clusters bridged by organic linking groups. Some MOFs exhibit extreme surface areas because of their porosities and exceptional capacities for storing gas molecules.
MOFs are especially attractive candidate filter materials because, in principle, scientists can build desired sorption properties into the crystals by pairing select metal and organic building blocks. Then they can further tune those properties by modifying the MOF with reactive functional groups. To identify the most effective functional group, researchers synthesize series of related MOFs with a variety of groups and iteratively evaluate the effects of functionalization on gas uptake.
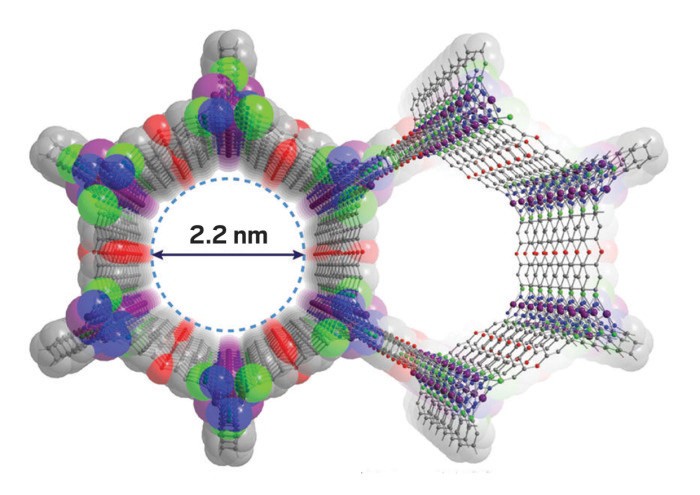
That’s an approach taken by chemical engineer Krista S. Walton of Georgia Institute of Technology. In a recent study, Walton and coworkers evaluated uptake of ammonia, a toxic gas used on an industrial scale, in a zirconium-based MOF known as UiO-66 and six of its variants (Chem. Eng. Sci. 2014, DOI: 10.1016/j.ces.2014.08.050). The series included UiO-66 functionalized with one or more OH, NO2, NH2, SO3H, and COOH groups. The study was conducted together with Peterson; Jared B. DeCoste, a research chemist with defense contractor Leidos; and others.
The team expected that the MOFs functionalized with the acidic SO3H and COOH groups would do the best job of filtering ammonia-laced air. But UiO-66-OH and UiO-66-NH2 came out on top instead. Walton thinks this is because the bulky SO3H and COOH groups restrict ammonia’s access to the MOF pores, where adsorption occurs. In contrast, OH groups, which interact strongly with ammonia, are the least bulky of the series and do not hamper access.
If pores are too small or blocked, toxic gas molecules cannot reach adsorption sites, Walton says. If they are too large, there may be insufficient interaction between the pore walls and the gas molecules for optimal trapping. “You need to find the sweet spot,” she says.
A handful of research groups have conducted similar systematic studies detailing the effectiveness of various MOFs at trapping poisons such as carbon monoxide, nitrogen oxides, sulfur dioxide, and other toxic compounds. These kinds of studies, which Peterson and DeCoste recently reviewed in Chemical Reviews, have helped point scientists to a set of guiding principles for preparing the most effective MOFs (2014, DOI: 10.1021/cr4006473).
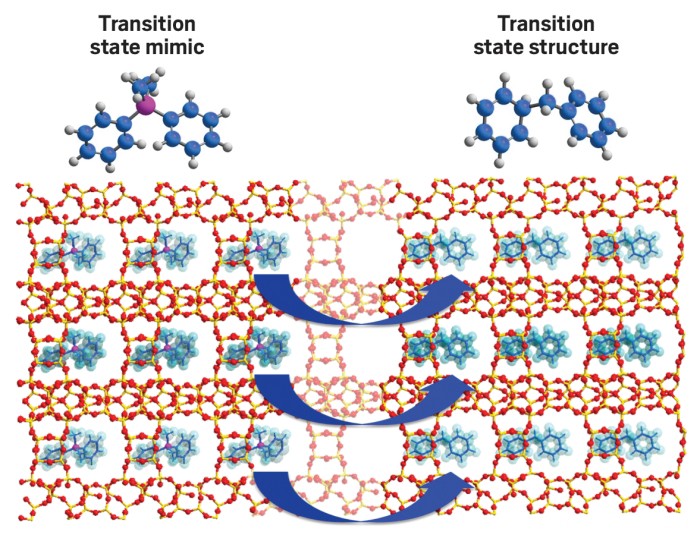
In addition to trapping toxic gases, some MOFs can neutralize the poisons chemically. For example, Northwestern University chemists Omar K. Farha and Joseph T. Hupp and coworkers noticed that Zr-OH-Zr linkages in UiO-66 resemble the Zn-OH-Zn active site in phosphotriesterase enzymes. These naturally occurring enzymes hydrolyze phosphate ester bonds. So Farha and Hupp wondered whether the MOFs could catalyze the same reaction in highly toxic organophosphate nerve agents, such as sarin and soman, rendering them harmless.
For the sake of safety, the team studied the less toxic simulants methyl paraoxon (dimethyl 4-nitrophenyl phosphate) and p-nitrophenyl diphenyl phosphate in solution phase. They found that the MOF effectively and selectively mediates cleavage of the simulants’ phosphate ester bonds (Angew. Chem. Int. Ed. 2014, DOI: 10.1002/anie.201307520).
“With nerve agents, cleaving the right bond is critical,” Farha says. “If you cleave the wrong bond, the product can be as nasty as the real thing.” Farha notes that the team is now working on new types of MOFs that appear to be effective at neutralizing real nerve agents, not just simulants, under more realistic gas mask conditions.

Ultimately, the broadest range of respiratory protection will probably be provided by combining materials, Peterson says. One promising non-MOF material undergoing extensive evaluation is a zirconium hydroxide sorbent doped with cobalt, zinc, silver, and amines. Known as CoZZAT, the material is effective at filtering acids and acid-forming gases such as cyanogen chloride, hydrogen cyanide, hydrogen chloride, hydrogen sulfide, and others. Unlike activated carbon, which traps poisons via relatively weak physical adsorption, CoZZAT filters these molecules by reacting with them chemically.
Combining CoZZAT with a MOF such as Cu-BTC, a Cu-based framework compound that effectively traps ammonia, amines, and related compounds, and with activated carbon, which readily adsorbs a wide range of organic compounds, would protect against a broad spectrum of poisonous gases. ECBC scientists are currently assessing the efficacy of such combinations in laboratory studies.
But materials that do well in the lab are only halfway home. “I’ve seen many technologies that work well in the lab but don’t survive in the harsh field environment,” says William M. Fritch, an ECBC product manager responsible for evaluating gas mask technology for the U.S. Army, Navy, Marine Corps, and Air Force. Filter materials (and the masks themselves) must pass a battery of tests that expose the equipment to high and low temperatures, temperature swings, high levels of humidity and dust, rough handling, and other real-world conditions, Fritch says. MOFs, used alone or in combination with other materials, have not yet been subjected to those elaborate field tests.
Whether MOFs will make it to that stage of gas mask development remains to be seen. Peterson and DeCoste are optimistic, especially because of the level of chemical customization possible with MOFs. But thus far, only a limited number of studies have been conducted, and most deal with just a handful of the most common framework compounds. To take advantage of the promise of MOFs, Peterson says, additional experts in materials synthesis and adsorption need to work together. Such collaborations could lead to advances that would make MOFs the stars of respiratory protection.










Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter