Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Gentle Exfoliation Yields Mof Membranes
by Mitch Jacoby
December 15, 2014
| A version of this story appeared in
Volume 92, Issue 50
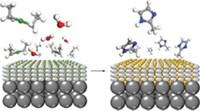
Thin sheets of metal-organic framework (MOF) compounds could be useful as gas-separating membranes. But researchers have had limited success in preparing these crystalline solids in two-dimensional form. Yanshuo Li and Weishen Yang of China’s Dalian Institute of Chemical Physics and coworkers have now demonstrated that MOFs with a layered structure can be exfoliated in a way that exposes 1-nm-thick sheets with large lateral dimensions and a high degree of crystallinity (Science 2014, DOI: 10.1126/science.1254227). MOFs are porous crystalline materials composed of metal ions or metal clusters bridged by organic linking groups. Some MOFs exhibit exceptional surface areas and gas storage and separation properties because of their elaborate system of pores and internal channels. For those applications, MOFs are typically used as powders. Easily processed membranes would be more convenient, but common methods for preparing membranes often damage the MOF structure, which is key to gas permeation. So Li, Yang, and coworkers devised a gentle exfoliation method that includes low-speed wet ball milling and ultrasonicating in a methanol-propanol mixture. In tests with hydrogen-CO2 mixtures, the team showed that a membrane formed from exfoliated sheets of a Zn-benzimidazole MOF efficiently separates the gases, allowing hydrogen to permeate easily and selectively.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter