Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
New Route To Improved Heparins
Drug Discovery: Chemoenzymatic analogs may sidestep key problems of current anticoagulants
by Stu Borman
March 3, 2014
| A version of this story appeared in
Volume 92, Issue 9

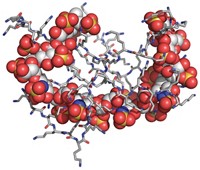
Researchers have used chemoenzymatic synthesis to create new forms of heparin that may solve key problems with current versions of the medication. Heparin, a sugar-based polymer, is widely used to reduce blood clotting in patients who have thrombosis and other conditions.
Two of the three commercial forms of heparin—unfractionated heparin and low-molecular-weight heparin—are derived from pig intestines. That means they are inhomogeneous mixtures of sulfated carbohydrates of different sizes and variable sulfation patterns, leaving them open to batch-to-batch variations and uncertain efficacies. Their animal source also makes them subject to virus and prion impurities and other types of contamination, some of which have caused patient deaths.
The third form, a synthetic pentasaccharide called fondaparinux, is a pure substance. But its effects are irreversible: It lacks an antidote when anticoagulant activity becomes excessive. Fondaparinux is primarily excreted through the kidneys, giving it long persistence in patients with kidney disease, for whom it is therefore unsuitable. Furthermore, it is difficult to synthesize, requiring 50 steps.
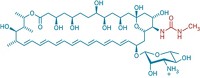
Robert J. Linhardt of Rensselaer Polytechnic Institute; Jian Liu of the University of North Carolina, Chapel Hill; and coworkers have now used chemoenzymatic synthesis to develop new heparin variants (Nat. Chem. Biol. 2014, DOI: 10.1038/nchembio.1459).
Up to now, the ability to develop improved heparin therapeutics “has largely been hindered by an inability to synthesize these complex molecules,” Liu says. “Our new chemoenzymatic method opens the possibility to expand heparin-based medicines for a wide variety of clinical indications.”
The most promising new heparin variant they synthesized is a dodecasaccharide (12-unit sugar). It is a pure synthetic substance like fondaparinux and therefore addresses the inhomogeneity problems of porcine heparins. It may be possible to administer it to kidney-impaired patients because it appears to be cleared, at least in part, through the liver. Its effects can be reversed by coadministering an approved drug called protamine. And it’s easier to synthesize than fondaparinux, requiring 22 steps.
Although heparin is most frequently used as an anticoagulant, it may also have anticancer, anti-inflammatory, and antiviral activities. The chemoenzymatic synthesis can be fine-tuned to enhance different heparin activities, Linhardt says. It might also make it possible to synthesize heparins that are easier to administer, last longer, and are optimally cleared from the body.
Jeffrey I. Weitz, a specialist in hematology and thromboembolism at McMaster University, in Ontario, comments that the new agent’s “potential for complete protamine reversal may render chemosynthetic low-molecular-weight heparins better choices than enoxaparin,” a widely prescribed porcine heparin, “for treatment of patients at high risk for bleeding, such as those who have undergone recent surgery.”
If its liver excretion properties are confirmed, the new heparin could also be useful in patients with severe renal impairment, for whom enoxaparin and fondaparinux cannot be used, Weitz adds. “This would be an important advance,” he notes. But he cautions that its ease of administration, anticoagulant activity over time, mode of excretion, and potential to lower the amount of blood platelets, a serious complication of heparin therapy, still need to be evaluated.
Liu says the team plans to determine whether large-scale synthesis of the chemosynthetic agents is feasible and hopes “to license the technology to a business partner for drug development.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter