Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Petrochemicals
Better Chemistry Flows To The Oil And Gas Industry
From fracking to deep sea drilling, ensuring high production and environmental protection requires a careful selection of chemicals
by Melody M. Bomgardner
April 13, 2015
| A version of this story appeared in
Volume 93, Issue 15

The boom in U.S. oil and gas production has brought huge economic opportunities for the country and for the chemical industry. But it has also brought close scrutiny of the chemicals used in processes such as hydraulic fracturing, also known as fracking. Between the economic hype and environmental backlash is the story of how chemicals are chosen and marketed for this industry.
It’s a story complicated by the nature of the supply chain and companies’ insistence on keeping their formulations proprietary. Chemical firms, oil-field service companies, and well operators are all involved in developing chemical recipes for use in oil and gas drilling. Many of the chemicals used, such as guar gum and ethylene glycol, are common and familiar, whereas the identities of others, such as surfactants and specialty polymers, are closely guarded trade secrets.
Still, the trends driving the development and selection of chemicals used in the field are clear. Oil and gas well operators are looking for chemicals that will help them economically produce more hydrocarbons from a single drilling site, reuse water from hydraulic fracturing, and reduce risks to the environment—roughly in that order of concern.
Specialty chemical firms have always served all kinds of oil and gas operations. These days, the rise of production from unconventional sources—shale rock, deep ocean reserves, and previously tapped-out oil wells—is driving demand for more sophisticated chemistry and an increased understanding of chemical interactions deep within geologic formations.

Source: Colorado Oil & Gas Conservation Commission
To capture a larger share of the growing industry, many chemical firms are expanding their offerings. In 2013, for example, Solvay acquired U.S.-based Chemlogics for $1.3 billion in cash. For Solvay and competitors such as Dow Chemical and Innospec, tricky performance requirements plus the sheer amount of oil and gas activity combine to make for a good business.
“The industry is changing,” says Norm Byrne, director of R&D for Dow’s oil, gas, and mining business. “Things are getting deeper—for example with deepwater drilling—and also getting hotter. Environmental compliance and regulatory issues are key for us, and we’re seeing a lot of pressure on increasing production and maximizing operations. What we’re seeing is things are becoming more complex.”
For instance, last month the Department of the Interior issued a rule that requires companies drilling on public lands to disclose the chemicals they use to the Bureau of Land Management. This and other calls for transparency may benefit the chemical industry, according to Chen Pu, Solvay Novecare’s executive vice president for oil and gas. The trend will help Solvay in the long run as the firm unveils more environmentally friendly chemicals for well operators, Pu says.
Disclosed or not, the substances used to get oil and gas out of the ground have many people concerned. For example, in hydraulic fracturing, several hundred different chemicals may be added to water used to open up fractures in shale rock and keep oil and gas flowing from horizontal drilling wells that can reach 5,000 feet in length. Because a single vertical drilling site can launch more than 20 horizontal wells, the amount of fluid used in fracking can be 20 times that of conventional wells.
And the diversity of substances commonly used in hydraulic fracturing fluids is difficult to overstate. Large-volume products include fairly benign ones, such as the plant-derived polymer guar, as well as more hazardous chemicals such as benzene and xylene. On the specialty ingredient side are polymers that biodegrade and leave little or no residue, as well as microbicides that biodegrade over time but are acutely toxic to aquatic life if they get into surface waters.
Chemicals make up less than 1% of the fluid that well operators use to fracture shale and release oil and gas; the rest is water and proppants, usually specialized sand, that hold the fissures open. The chemicals include thickeners and friction reducers to help deliver the proppants way out in the horizontal wells. Other additives are used to break down the thickened goo, prevent bacteria growth, and protect valuable equipment from corrosion.
After the fracking process is complete, the water that comes back up from the wells still contains most of those chemicals. This so-called produced water also contains salts and minerals from rock formations and must be treated or reused; spills into soil or water are a problem. The environmental fate of the chemicals and breakdown products from fracking is an active area of research and is important to understanding overall risk (C&EN, March 16, page 8).

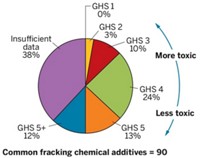
Much of the publicly available information on chemicals used in hydraulic fracturing comes from www.fracfocus.org, a voluntary data repository where well operators report their chemical use. An early-2013 analysis of the website for the environmental group Clean Production Action (CPA) assessed to what degree hazardous chemicals were associated with hydraulic fracturing. Data from 6,500 wells in 20 states turned up CAS Registry Numbers indicating 650 unique chemicals, according to the report’s author, Brian Penttila, a chemical engineer at the consulting firm iWatchChems in Seattle.
The initial analysis showed 96 chemicals that, according to CPA codirector Lauren Heine, are of very high concern and 132 where the level of hazard is unknown. Heine says CPA has put 40 of the most commonly used chemicals through its hazard assessment program, called GreenScreen. The list includes methenamine, a corrosion inhibitor; ethylene glycol, used as a cross-linker and friction reducer; and a nonylphenol ethoxylate. CPA has made the reports available on its website.
One question that comes up, particularly in hydraulic fracturing, is how much the well operators themselves know about what is in their fracturing fluid. That’s because the chemicals may have been selected and sold as a package by an oil and gas service company such as Halliburton or Schlumberger. Neither company agreed to speak with C&EN for this story.
But business practices are changing and some operators are buying direct, says Danny Durham, a chemist who directs upstream chemical operations at Apache, a Houston-based oil and gas exploration and production company. “Operators are hiring people like me from the chemical side of the business to source specific chemicals based on cost, performance, and sustainability.”
Durham says Apache decided to bypass the oil-field service firms on chemicals for reasons of cost and sustainability. Whereas service companies may charge more for “green” fracking products, Durham says he can identify chemicals that meet sustainability guidelines and are cheaper than what are normally used. “For example, Apache has sourced a friction reducer at a cost cheaper than what most people source, and it is drinking-water-approved” by the Environmental Protection Agency, he says.
No industry-wide criteria exist for what makes a “green” fracking recipe; instead, each oil and gas service company sets its own bar for sustainability. For example, on its website, Halliburton touts its use of substances generally recognized as safe by the Food & Drug Administration for use in food. Chemicals that are biobased and/or biodegradable are often called upon to replace longer-lasting synthetics. Water-based ingredients are favored over oil-based ones.
For its part, Apache tries to choose substances that meet the standards of EPA’s Design for the Environment (DfE) program. “Over the years, if you pay attention, you figure out how you can pick out the right products,” Durham says. “I start with sustainability as my goal. It turns out that if I want a product line that is EPA DfE listed by CAS Registry Number, there is quite an extensive list to choose from.”
As the chair of the Green Chemistry Institute’s recently established Hydraulic Fracturing Roundtable, Durham will have the opportunity to share his expertise and gain new insights from others in the industry. GCI is a division of the American Chemical Society, which publishes C&EN.
The roundtable will examine the current slate of chemicals used in hydraulic fracturing, including solvents, microbicides, and corrosion inhibitors, and work to develop more sustainable alternatives. According to Durham, the main concern is the risk of surface spills; chemicals in the well generally break down during the life of the well’s operation, which can be years.
Spills do happen, though they appear to be rare. Colorado is home to more than 53,000 active oil and gas wells, many of them along the front range of the Rocky Mountains, a corridor that is also home to the residents of Denver, Boulder, and Fort Collins. In 2013, 600 spills at well sites were reported to the Colorado Oil & Gas Conservation Commission, up from 399 in 2012. The volume of water spilled was a mere 0.004% of the total produced water from all wells.
According to the commission, leading causes of spills at oil and gas wells are equipment failure and human error. Of the 399 spills reported in 2012, 63 impacted groundwater and 22 impacted surface water. The commission does not track spills that happen during the transportation of chemicals or produced water.
One environmental strategy that the industry has taken up over the past two years is to reuse the water produced by one well in a nearby well, according to A. Daniel Hill, professor of petroleum engineering at Texas A&M University. But the salts and minerals in produced water require changes to the chemical routine.
For example, chemicals used to thicken fracking fluids so they can carry proppants far distances underground might not gel properly in salty water. And the poor-quality water may require new additives such as cross-linkers, Hill says. Cross-linkers—traditionally borate salts and zirconium complexes—are used to firm the gel formed from guar gum and other thickeners.
What’s more, after the proppants are set up in the fractures, operators require that the viscous mixture reliquefy so the oil and gas can flow out. So they add special time-release breakers to make the gel fall apart. To ensure this happens in salty water, Texas A&M researchers are investigating ways to encapsulate breakers, including nano-entrapped structures, Hill reports.
At Dow, researchers are looking for ways to make oil and gas wells more productive even as the down-hole environment becomes more extreme. Salinity and high temperatures and pressures are all increasingly common, Byrne says. Thus, Dow’s scientists work in labs where they can simulate being 2 or 3 miles below the earth or several thousand feet below the ocean.
“Part of what we offer today are rheology modifiers, such as cellulosic ones, and products able to withstand high temperatures,” Byrne explains. Rheology modifiers let operators tune the viscosity of the water-chemical mixture that moves proppants. “These are ‘triggerable’ depending on pH, temperature, and time,” he says. “That way you can get them to act when and how you want them to.”
Although the conditions down the well are challenging for chemists, they are surprisingly nurturing for microbes. Bacterial populations, whether they originate from the fracking water or from deep in rock formations, multiply exponentially when served a huge meal of biopolymers. Particularly problematic are sulfate-reducing bacteria, which emit corrosive and toxic hydrogen sulfide.
“Microbes cause major problems including corrosion of equipment and souring of the well—which is when the bacteria plug the flow through the well and impact production,” says Rick Strittmatter, R&D director for Dow’s microbial control business. The small but mighty bugs can negatively impact both the quality and quantity of oil and gas from the well. By using microbicides, “folks are realizing they can get more out of their existing wells, and that’s more sustainable,” Strittmatter says.
One of Dow’s leading microbicides, glutaraldehyde, is a favorite among those formulating low-toxic fracking fluids. Although it is classified as acutely toxic and requires safe-handling procedures similar to bleach, glutaraldehyde has a fan in Apache’s Durham because “it has very little chronic toxicity and fares very well in bioaccumulation and biodegradation testing.”

For a PDF of this graphic click here.
In fact, the aqueous biocide is so popular that Dow reports counterfeit products are common. The fake stuff is usually a combination of formaldehyde, glyoxal, and other chemicals and often comes from China.
Advertisement
At Solvay, one way to reduce potential environmental side effects from biocides and other chemicals is to combine them with a biodegradable polymer. This system can cut the amount of active chemicals required by 20% and helps operators improve the environmental profile of their wells by allowing them to reuse water multiple times. “Polymer chemistry is really the future,” Pu says.
Polymer chemistry also comes into play in the area of viscosity control. Pu says he benefits from Solvay’s deep knowledge of specialty polymers such as cross-linkable compounds, which are used in a variety of products.
An example with large-scale application to oil and gas is derivatized guar gum, a chemically modified polysaccharide also used in hair conditioners and as an antidrift agent for agricultural chemicals. The strength added to the guar means operators can get proppant to the farthest reaches of the horizontal well.
In some cases, however, use of large amounts of a strong thickener can affect the delicate structure of the proppant dispersion. Independence Oilfield Chemicals, which is part of Innospec, has developed a low-guar recipe with an added specialty cross-linker. The company says the product works well to deliver proppants and leave behind an intact structure because it is both viscous and elastic.
As a specialty chemical maker, Innospec has bought regional suppliers to the oil and gas business such as Houston-based Independence and Oklahoma City’s Bachman Services. Its goal, according to Brian R. Watt, vice president of strategic planning, is to provide a custom chemical service directly to the well operator. “We’re working to roll that service model out and continue to put research and development dollars into new products that are either better performing or more environmentally friendly,” he says.
If companies with new chemical offerings can sell directly to oil- and gas-field operators, the door may open for younger firms to enter an industry that has been difficult to access. One start-up, called Clean Chemistry, now claims to have its first customers for an on-site water-reuse system that relies on a reactive oxygen species.
“We identified an opportunity to get to market quickly and with less technical risk by commercializing a chemistry that we synthesize with standard merchant chemicals,” explains company cofounder Damon Waters. The reactive oxygen product only lasts for a few hours—which is why it is created on-site—but it requires very little hardware, according to Waters. Once made and added to the wastewater, it can replace other chemicals to control bacteria, suspended metals, and dissolved solids, enabling reuse.
Biobased chemical firms also have identified the oil and gas industry as a potential customer for their products. For example, Elevance Renewable Sciences, which makes vegetable-oil-derived ingredients for cleaning and personal care products, says its Clean 1200 product works well as a solvent in place of hydrocarbons such as benzene.
According to Frederyk Ngantung, head of technical marketing for Elevance, the bioderived cleaner removes paraffin wax deposits that block the flow of oil and gas wells. In addition to delivering performance, he says, it is more environmentally friendly, offering low volatility and flammability and ready biodegradability.
Owing to the current dip in energy prices, it’s difficult to get oil and gas operators to replace familiar chemistry with something new, Solvay’s Pu acknowledges. “Once the market stabilizes, in the next 12 to 24 months, the trend of removing many of the nasty components will accelerate again; it’s a trend that has already started.”
As evidence, Pu points to drillers in some areas of the North Atlantic that operate under stringent chemical restrictions mandated by the Convention for the Protection of the Marine Environment of the North-East Atlantic, or OSPAR, for more than two decades.
In 2010, OSPAR reported that discharges of substances viewed as high priorities for control had been reduced by 90% since 2003, mostly through substitution by less hazardous alternatives.
In the North Atlantic and in other oil and gas regions, exploration and production firms have drastically cut back on drilling new wells. “There will be pressure on operators to get a lot more production on the wells they have or are going to drill,” Innospec’s Watt says.
That could turn out well for firms that have the chemical expertise to help operators contain costs and limit environmental impact while optimizing their output. “You can have your cake and eat it too,” Durham says, “but you need to have someone knowledgeable to get you there.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter