Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Physical Chemistry
Iron Oxides Exhibit Greater Complexity
Geochemistry: Redox chemistry in Earth’s interior may be more complex than thought
by Elizabeth K. Wilson
June 29, 2015
| A version of this story appeared in
Volume 93, Issue 26
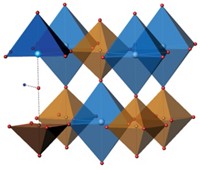
Researchers have synthesized a new iron oxide, Fe5O6, adding to evidence that the two most abundant elements on Earth form a complex chemical system that has been previously unrecognized (Sci. Adv. 2015, DOI: 10.1126/sciadv.1400260). Iron oxides play a major role in the redox chemistry of rocks, particularly deep in the interior of Earth, and possibly other planets. Until recently, scientists believed iron oxides came in three forms: FeO, Fe2O3, and Fe3O4. In 2011, Barbara Lavina at the University of Nevada, Las Vegas, and Yue Meng at the Carnegie Institution of Washington, Argonne, Ill., upended that belief with their synthesis of Fe4O5 (Proc. Natl. Acad. Sci. USA, 2011, DOI: 10.1073/pnas.1107573108). Lavina and Meng have now identified Fe5O6 from mixtures of iron and hematite, which they heated and pressurized in the laboratory. “Fe5O6 and Fe4O5 are plausible new players in Earth’s mantle redox equilibria,” the authors note.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter