Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Pairing Two Catalysts Promotes Synthesis Of Unsymmetrical Biaryl Compounds
Cross-Coupling: Cooperative nickel and palladium complexes overcome the challenge of connecting two different aryl electrophiles
by Stephen K. Ritter
August 21, 2015
| A version of this story appeared in
Volume 93, Issue 33

In a surprising twist on classical cross-coupling chemistry, a research team at the University of Rochester has discovered the first general method for making unsymmetrical biaryl compounds directly from two different aryl electrophiles.
The new approach, which involves using two metal catalysts to promote the aryl-aryl coupling of unwilling partners, is being hailed as a means of simplifying the synthesis of complex molecules such as pharmaceuticals. The approach also provides new insight into the selective transfer of ligands between two transition-metal catalysts, a strategy that can be applied to a range of chemical reactions (Nature 2015, DOI: 10.1038/nature14676).
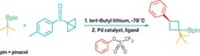
The work by Rochester’s Laura K. G. Ackerman, Matthew M. Lovell, and Daniel J. Weix is a variation on the century-old Ullmann reaction for coupling an aryl halide with itself. Extending the reaction to efficient cross-coupling of different aryl halides with a single metal catalyst has remained challenging, Weix notes.
Weix’s group found that pairing two catalysts with similar reactivity but different preferences for aryl electrophiles could streamline the reaction. The researchers used a nickel bipyridine catalyst that reacts with aryl bromides to form a transient intermediate combined with a palladium phosphine catalyst that reacts with aryl triflates to generate a persistent intermediate.
The team hasn’t pinned down the exact mechanism, but the two intermediates exchange ligands followed by a reductive elimination step at one of the metals to form the biaryl product. Although the catalysts form less than 5% of the unsymmetrical biaryl product on their own, together they achieve up to 94% yield.
The new paper is “fantastic” work, says UCLA’s Neil K. Garg, whose group specializes in cross-couplings. “Cross-electrophile couplings have been building momentum, especially because of the many contributions of the Weix group. This breakthrough methodology is certain to see extensive use in academia and industry.”
Catalysis expert Shannon S. Stahl of the University of Wisconsin, Madison, agrees. “It seems ridiculous to say this, given the long history of cross-coupling and the advances made in recent years, but the Weix group’s approach could significantly expand the scope of cross-coupling chemistry.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter