Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Web: Targeting Protein Garbage Disposal To Treat Alzheimer’s
Neuroscience: Small molecule keeps protein-degrading proteasomes active in mouse brains
by Michael Torrice
January 4, 2016
| A version of this story appeared in
Volume 94, Issue 1
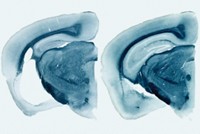
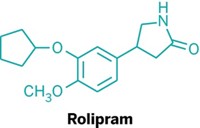
In neurodegenerative diseases, certain proteins misfold and clump in the brain. A study of mice reports that aggregated tau—a protein associated with Alzheimer’s disease—can impair the function of cells’ proteasomes, garbage disposals that cells use to break down misfolded proteins. Ramping up the activity of proteasomes with a small molecule overcame this impairment, leading to fewer tau aggregates and improved cognitive function. Natura Myeku of Columbia University and colleagues studied genetically engineered mice that express a tau protein prone to aggregate. Proteasomes isolated from these mice had less protein-dicing activity than those from normal animals. The team thinks that tau gloms on to the complex and disrupts its function. To help proteasomes overcome this disruption, the researchers turned to rolipram, a compound that triggers a pathway that phosphorylates the proteasome, a mechanism known to increase proteasome activity. When given to four- to five-month-old engineered mice, the molecule led to more active proteasomes, fewer tau aggregates, and improved spatial memory, compared with control mice (Nat. Med. 2015, DOI: 10.1038/nm.4011).


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter