Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Business
Indian drug firms struggle with quality issues
More and more companies are challenged by FDA’s scrutiny of manufacturing records
by Jean-François Tremblay
April 18, 2016
| A version of this story appeared in
Volume 94, Issue 16

When visiting drug plants outside their home country, U.S. Food & Drug Administration inspectors typically have just a few days to determine whether the facilities comply with manufacturing standards. In the case of Pan Drugs, inspected in July 2014, it was an easy task.
“Our investigator observed holes in the walls and roof which allowed pigeons access near production equipment in multiple manufacturing areas,” read the warning letter that FDA sent to the firm last September to explain why products made at the plant, in Vadodara, India, would be banned from the U.S.
Dr. Reddy’s Laboratories, one of the biggest names in India’s drug industry, similarly failed spectacularly. Several days into a late-2014 inspection, FDA officials discovered a lab, not previously disclosed by the firm, that had analyzed active pharmaceutical ingredients exported to the U.S. Inspectors found that the secret lab kept retesting batches that had failed quality tests until positive results were obtained.
“Until May 2013, regulators in the U.S. and Europe took what was presented to them by companies based in India at face value.”
-Dinesh S. Thakur, executive chairman, Medassure Global Compliance
More often, though, the problems that FDA uncovers are less obvious than they were at Pan Drugs and Dr. Reddy’s. In recent years, dozens of plants in India have been cited for “data integrity” issues in warning letters sent by FDA officials or their regulatory counterparts in other countries. Harder to fix than holes in a wall, data issues are likely to plague Indian firms for years to come and could threaten their role as leading U.S. suppliers of generic drugs.
“ ‘Data documentation issues’ is not the case of a sloppy secretary failing to file test reports alphabetically but a serious issue, one which has an impact on the lives of human beings,” wrote Dinesh S. Thakur last month in the Indian magazine Business Today. “If a batch fails a test, a manufacturer is required to withdraw the consignment from the market—in most cases this would result in a loss to the manufacturer. The simpler, unethical, and illegal way around this problem is to delete the failed test records and replace them with manipulated test records that demonstrate that the batch passed quality assurance tests.”
Based in Tampa, Fla., Thakur is head of Medassure Global Compliance, a consulting firm advising companies on their pharmaceutical supply chains. Thakur was the whistle-blower who alerted FDA in 2007 to systematic manipulation of manufacturing records at Ranbaxy Laboratories. The company was fined $500 million in 2013 for its transgressions. Thakur was expected to receive about 10% of it.
It was the fraud committed by Ranbaxy, which is now part of Sun Pharmaceutical Industries, that led FDA to more broadly question the validity of data submitted by drug manufacturers, Thakur tells C&EN. “Until May 2013, regulators in the U.S. and Europe took what was presented to them by companies based in India at face value,” he says. “Ranbaxy changed that.”

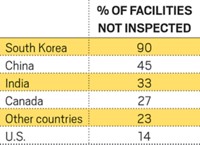
Although FDA’s new focus on data integrity is worldwide, it has proven most damaging to India. In the past five years, of the 29 warning letters citing data integrity issues that FDA sent to companies around the world, 18 related to facilities in India (C&EN, April 4, pg. 15). A common problem uncovered in its inspections is failure to prevent unauthorized access to test data, which leaves inspectors unsure as to whether the data companies are presenting amount to a complete and accurate picture of their manufacturing performance.
Thakur reckons that 44 Indian drug plants are currently banned from exporting to the U.S., a number that he says is evidence of a systematic problem with the country’s drug industry.
According to a recent report by the Indian Pharmaceutical Alliance, an industry group whose members include India’s most prominent drug firms, a total of 379 plants have at some point been authorized by FDA to export pharmaceuticals to the U.S. Fully a third of the drugs sold in the U.S. in 2014 were manufactured in India, the report says.
The focus on data integrity at FDA, and at its counterparts in Europe and Japan, has kept Indian firms on alert for some time. Yusuf K. Hamied, the chairman of Cipla, which has yet to receive an FDA warning letter, told C&EN in December 2013 that whenever a competitor receives such a letter, Cipla employees review it to make sure that the firm’s facilities don’t have a similar problem.
At the time of the interview, a manufacturing quality consultant who previously worked at the World Health Organization was auditing one of Cipla’s main active ingredient plants to make sure its systems were in order.
That kind of self-auditing is costly, but failure to meet FDA requirements is likely dearer. In 2013, for example, the agency issued two warning letters to Wockhardt, banning two of the company’s plants from exporting to the U.S.
Until then, Wockhardt’s sales in the U.S. had been steadily expanding, to the point that they accounted for more than 40% of total sales. With two of its plants banned from exporting to the U.S., Wockhardt’s sales dropped by 14% in the fiscal year that ended March 31, 2014. Profits fell by nearly half.
Given that the financial penalty for noncompliance can be so high, it is perplexing that many Indian drug exporters have not taken the steps required to ensure that their manufacturing standards meet those of Western regulators. But thoroughly fixing the issues could harm the business models of many of them, claims Peter Saxon, president of Saxon International Associates, a New Jersey-based consulting firm that has advised dozens of Indian and Chinese drug companies on meeting U.S. drug manufacturing regulations.
“India has always been admired for getting products to market fast,” Saxon says. When a drug goes off patent, the first company to come up with a generic version is often an Indian firm. But in rushing, he claims, companies risk going to market with manufacturing processes that do not consistently produce quality drugs.
Quality control managers at these firms end up with the job of falsifying data to cover up their poorly designed manufacturing processes, Saxon says.
According to Thakur, the Ranbaxy whistle-blower, such data falsification comes easily to Indian firms because Indian regulators are much less demanding than Western ones. “Indian pharma companies have thrived in a lax and corrupt regulatory environment for over 30 years now and are not going to change their ways overnight,” he says. Regulators in India do not even investigate companies that have been banned from Western countries, Thakur points out.
The West shares some of the blame, according to drug safety experts. In the U.S., the focus has long been more on drug cost containment than on positive patient outcomes, Thakur says.
One result is that the quality regulation of generics—which account for almost 90% of the drugs sold in the U.S.—is more lax than that of patented drugs, Saxon notes.
In view of the prevalence of quality problems at drugs plants in India and elsewhere overseas, consumers should know where their drugs—and the active ingredients they contain—are made, argues Helena Champion, principal consultant at Drug Quality Assurance, a Boston-based drug manufacturing quality and regulatory compliance firm.
“Consumers can read on the label where a clothing item is made, but not where a drug is made,” she says. “Companies can market under a known U.S. or European name and address, even though the product may actually be made by another company in a country where fraud prevails.”
Although it will likely take years for Indian drug manufacturers to solve their data integrity issues, a consensus is emerging within India that the country’s pharmaceutical industry needs to change its ways.
Assocham, a federation of Indian chambers of commerce, urged the Indian government in December to tighten its regulation of the drug industry to force companies to raise their standards and compete on quality. Meanwhile, the Indian Pharmaceutical Alliance has set up a quality forum, one of the main thrusts of which will be to help its members establish robust data and documentation systems. Several alliance members have received warning letters from FDA.
Despite the Indian drug industry’s problems, the credit rating agency ICRA is optimistic about prospects for Indian firms in the U.S. market. In a report last month, ICRA noted that growth at several leading companies had been curtailed by regulatory issues. But on the whole, it expects the industry to continue launching generic drugs in the U.S. soon after their original versions lose patent protection.
Thakur, the consultant, isn’t so sanguine. Regulators, patients, and buyers in the U.S. are catching on to India’s quality shortcomings, he says. And as medical evidence of the impact of substandard pharmaceuticals grows, he expects an American backlash against Indian-manufactured drugs.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter