Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Triple adamantane expands the boundary of phosphine ligands
Trisubstituted phosphine sets a new standard for electron-releasing character in an alkane-based ligand
by Stephen K. Ritter
May 30, 2016
| A version of this story appeared in
Volume 94, Issue 22
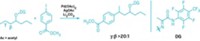
Phosphine ligands have played a supporting role in the success of many a transition-metal catalyst, helping to tune their activity, selectivity, and stability. Among the examples, tri(tert-butyl)phosphine has long been characterized as the most electron-releasing alkylphosphine ligand for metals. Liye Chen, Peng Ren, and Brad P. Carrow of Princeton University have succeeded in raising the bar with tri(1-adamantyl)phosphine, PAd3, a ligand that now provides a bridge between trialkylphosphines and the stronger electron-releasing N-heterocyclic carbene ligands (J. Am. Chem. Soc. 2016, DOI: 10.1021/jacs.6b03215). Adamantane is an unusual rigid C10 tricyclic alkane. Researchers have made phosphine ligands containing two adamantyl groups before, and adamantane is occasionally used as a bulky substituent for N-heterocyclic carbenes. But installing a third bulky adamantyl group on phosphorus has been a challenge, one that researchers took on with complex approaches. Carrow’s group pulled it off with a simple substitution reaction between commercially available HPAd2 and AdCO2CH3. The researchers demonstrated the utility of the surprisingly stable PAd3 by forming the palladium catalyst shown above and using it for Suzuki-Miyaura couplings, including using chlorinated heteroarenes to make drug intermediates and derivatives, with good results.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter