Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Busting a myth about mechanochemical crystallization
Adding varying amounts of liquid yields multiple crystal forms
by Jyllian Kemsley
August 1, 2016
| A version of this story appeared in
Volume 94, Issue 31
Although it may seem counterintuitive to put a compound into a ball mill to turn it into a crystalline form, the approach nonetheless works—and adding varying amounts of liquid can determine the crystal form that results, reports a team led by Bill Jones of the University of Cambridge (Cryst. Growth Des. 2016, DOI: 10.1021/acs.cgd.6b00682).

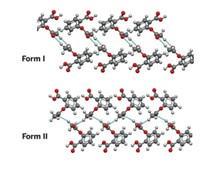
Compounds of interest for materials and pharmaceuticals applications often crystallize into different forms, called polymorphs. Because polymorphs can have varying stability, solubility, and other properties, forming a specific polymorph can be critically important.
Chemists have long thought that using one particular liquid when crystallizing compounds via mechanochemical milling always yields one particular polymorph. Seeking to test that dogma, Jones and coworkers crystallized 200 mg of a 1:1 equimolar mixture of caffeine and anthranilic acid using a ball mill, adding from 10 to 100 μL of 15 different liquids.
Four liquids—acetonitrile, nitromethane, ethylene glycol, and 1,6-hexanediol—formed one polymorph each, regardless of the amount of liquid. The rest of the liquids yielded different polymorphs or mixtures, depending on liquid volume: 10 to 20 μL of ethanol formed polymorph II, for example, whereas 40 to 60 μL formed polymorph I. Additionally, 10 μL of 1-hexanol, 1-octanol, or 1-dodecanol formed polymorph III, a polymorph previously only prepared by desolvation.
Similar effects could occur for single-component crystals, the authors say. The mechanism behind the phenomenon remains to be determined; the authors suggest that it could be a result of thermodynamic stabilization of nanoparticles, different growth mechanisms of the polymorphs, or changes in the free-energy difference between polymorphs caused by milling conditions.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter