Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Gene Therapy
CRISPR edits sickle cell mutation
Edited blood stem cells could someday help patients produce healthy red blood cells
by Ryan Cross
October 12, 2016
| A version of this story appeared in
Volume 94, Issue 41

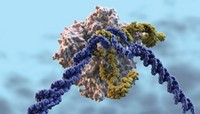
In a promising step toward a cure for sickle cell disease, researchers have used CRISPR/Cas9 gene editing technology to fix the genetic mutation underlying the condition in cells that eventually turn into red blood cells (Sci. Transl. Med. 2016, DOI: 10.1126/scitranslmed.aaf9336). The scientists envision that doctors could inject corrected cells into patients with the disease, allowing their bodies to make blood cells without the sickle cell mutation.
Sickle cell disease arises from a single mutation in the β-globin gene, which forms part of the hemoglobin protein that ferries oxygen throughout the body. The painful disease affects about 100,000 people in the U.S., mostly African Americans, and gets its name from the characteristic shape that red blood cells adopt due to polymerization of mutant hemoglobin. “Even with the best medical care, many people die in childhood,” says hematologist David I. K. Martin from Benioff Children’s Hospital, who was part of the study.
Jacob Corn from the University of California, Berkeley, who led the study, says that “we’ve theoretically known how to cure the disease since 1949.” That year, Linus Pauling popularized the term “molecular disease” after discovering sickle cells had mutant hemoglobin proteins. Later, it became clear that reverting a portion of the mutant gene, specifically a GTG sequence, back into the wild type sequence, GAG, should cure the disease.
In the new study, Corn and colleagues performed this reversion in hematopoietic stem/progenitor cells (HSPCs) obtained from the blood of sickle cell patients. Up to 25% of the mutant β-globin sites were fixed, and levels of wild type hemoglobin increased in edited cells, while levels of sickle hemoglobin decreased.
For gene editing to work as a long-term cure, edited HSPCs need to be incorporated into a patient’s bone marrow so the cells can serve as a source of healthy red blood cells. Some sickle cell patients undergo bone marrow transplants to cure the disease, but Martin says that option is an unpopular choice due to risks of complications such as graft rejection.
“Our hope with gene editing is to enable people to be their own bone marrow donors to reduce transplant risk,” Corn says. To test the concept, the researchers injected HSPCs into mice and found that the cells stuck around for at least 16 weeks, and at significant levels. Corn wants to find ways to boost the effectiveness of β-globin gene editing and long-term incorporation of edited HSPCs before considering clinical trials.
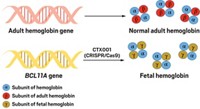
Hematologist Mitchell J. Weiss from St. Jude Children’s Research Hospital applauds the work. He led another team that recently reported using CRISPR/Cas9 to create a mutation in sickle cells that turns on production of fetal hemoglobin in adult cells (Nat. Med. 2016, DOI: 10.1038/nm.4170). Because fetal hemoglobin doesn’t contain the sickle cell mutation, this has been another long-sought strategy for curing the disease. “It is important to try every possibility because we don’t know what is going to work best,” Weiss says.
“Sickle is a ‘poster child’ for gene correction,” says Stuart H. Orkin a blood stem cell biologist at Harvard Medical School. “There is heightened interest given the hoopla around CRISPR/Cas9 but there are other ways to edit,” Orkin says, highlighting techniques such as gene therapy. “Ultimately, the approach that achieves excellent patient outcomes without side-effects is what is desired and there may be several different ways to achieve this end,” he says.
This article has been translated into Spanish by Divulgame.org and can be found here.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter