Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Cyclic peptides with heterocycles are cell membrane-permeable
Oxadiazole-containing peptidomimetic macrocycles have potential as drugs
by Stu Borman
October 31, 2016
| A version of this story appeared in
Volume 94, Issue 43
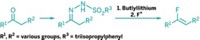
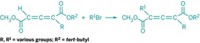
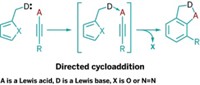
Peptides, including cyclic peptides, generally don’t cross cell membranes easily, a property that has limited their use in medicine. Researchers have now boosted the drug potential of cyclic peptides by incorporating a heterocycle into their rings. Andrei Yudin and coworkers at the University of Toronto adapted an existing reaction to convert linear peptides into the heterocycle-containing peptidomimetics (Nat. Chem. 2016, DOI: 10.1038/nchem.2636). The one-step reaction of a linear peptide with a variable aldehyde and (N-isocyanimino)triphenylphosphorane doesn’t require peptide preactivation, as most current peptide cyclization reactions do. Yudin and coworkers demonstrated the reaction by constructing 15-, 18-, 21-, and 24-membered peptidomimetic macrocycles, each containing 1,3,4-oxadiazole and an amine. The high cell-membrane permeability of many of the macrocycles and their conformationally rigid structures are both important prerequisites for bioavailability and therapeutic use. The researchers’ ultimate goal is to develop the peptidomimetics as inhibitors of intracellular protein-protein interactions. The phosphorane reagent “is being commercialized by Sigma-Aldrich, which will make this macrocyclization accessible to the chemistry community,” Yudin says.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter