Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Consumer Safety
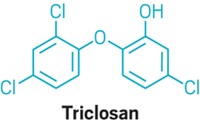
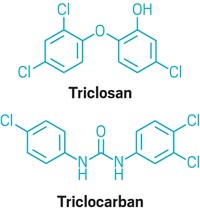
FDA bans triclosan and triclocarban in consumer soaps
Agency says manufacturers have not demonstrated safety or effectiveness of these antibacterial chemicals
by Britt E. Erickson
September 2, 2016

As of next year, companies will no longer be allowed to sell hand and body soaps in the U.S. that contain certain antibacterial ingredients, including triclosan and triclocarban.
In a final rule issued on Sept. 2, the Food & Drug Administration says that manufacturers have not shown that the chemicals are safe for long-term daily use. Companies also haven’t shown that the chemicals are more effective than washing with regular soap and water to prevent the spread of germs, FDA says.
“Some data suggests that antibacterial ingredients may do more harm than good over the long-term,” says Janet Woodcock, director of FDA’s Center for Drug Evaluation & Research. FDA proposed the rule in late 2013, citing concerns about potential hormonal effects and antibiotic resistance associated with the chemicals.
The rule goes into effect on Sept. 6, 2017, but many manufacturers have already stopped using the ingredients. As alternatives, most have switched to one of three other antibacterial chemicals: benzalkonium chloride, benzethonium chloride, and chloroxylenol. At the request of manufacturers, FDA deferred from including these three compounds in the new rule.
The American Cleaning Institute and its member companies plan to submit to FDA safety and effectiveness data for these three ingredients in the coming year, says Brian Sansoni, a vice president of the trade group. In the meantime, manufacturers can continue to market products that contain the three substances.
FDA’s action is part of a settlement made with the environmental group Natural Resources Defense Council. The group sued FDA in 2010 for not finalizing a 1978 rule that would have banned triclosan in consumer soaps.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter