Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Theoretical Chemistry
Density functional theory heads the wrong way
Theoretical method may be getting the right answer for the wrong reasons
by Mitch Jacoby
January 5, 2017
| A version of this story appeared in
Volume 95, Issue 2
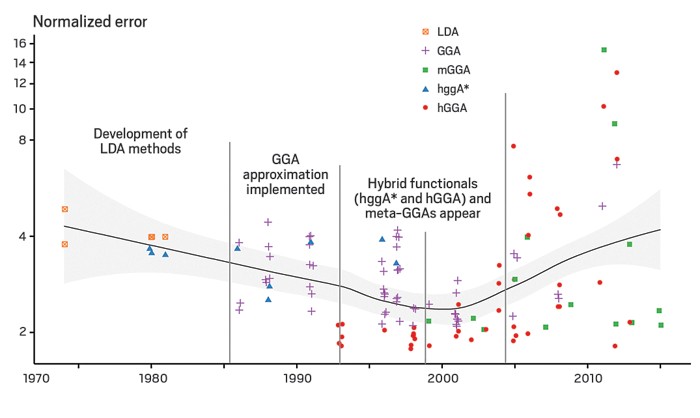
Density functional theory (DFT) is a widely used computational method for carrying out quantum calculations in chemistry, materials science, and biology research. Despite its enormous popularity and ongoing modifications and updates, DFT seems to be getting worse at predicting key electron properties, according to a study (Science 2017, DOI: 10.1126/science.aah5975).
The finding suggests that users of DFT programs should carefully evaluate and benchmark results of their computations. And it may encourage DFT specialists and developers to redouble their efforts to improve the method’s capabilities.
For decades, researchers have depended on quantum methods to calculate electronic structures, bond lengths, and molecular geometries and energies. The values of those properties and others can be obtained with high accuracy from wave-function-based quantum methods. But applying those methods to all but the simplest chemical systems is complex, arduous, and expensive.
DFT simplifies the calculations. It sidesteps the use of wave functions to account for the motions of a molecule’s atoms and electrons. Instead, DFT determines electronic properties from the three-dimensional densities of the systems’ electron clouds. That simplification has helped put quantum calculations in the hands of large numbers of researchers, not just hard-core theoreticians.
But as Michael G. Medvedev and Ivan S. Bushmarinov of the Russian Academy of Sciences and coworkers now report, although DFT continues to provide ever more accurate energy values, thanks to ongoing method development and refinements, it is getting worse at correctly predicting electron densities.
The team carried out DFT calculations on 14 types of atoms and ions using 128 different functionals—mathematical descriptions of electron density—developed since 1970. They compared those results with ones obtained from high-level ab-initio wave function quantum methods, which are known to be highly accurate. They find that until about 2000, DFT-calculated energies and electron density values improved hand-in-hand. Since then, however, the energies have continued to improve, but the densities have become less accurate.
For some applications in chemistry and biology, the energies and geometries of molecules are the most important pieces of information, says Sharon Hammes-Schiffer, a chemistry professor and specialist in computational methods at the University of Illinois, Urbana-Champaign.
“If the electron density does not affect these properties then perhaps the inaccurate electron density is irrelevant,” she adds. It may have little effect on chemical bonding.
The key issue in Hammes-Schiffer’s view is that some modern functionals “may be giving the correct energies for the wrong reason.” She argues that this subject merits further investigation because “most scientists would prefer to obtain the correct answer for the correct reason.”
Describing the study as “provocative,” Martin Head-Gordon, a theoretician at the University of California, Berkeley, says one way to move forward is a combinatorial approach to develop new functionals that use fewer adjustable parameters than some modern ones use. His group is working on that approach and they plan to test it on electron densities.
This article has been translated into Spanish by Divulgame.org and can be found here.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter