Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Tetratriflylpropene: A new organic superacid is in town
Chemists design a strong allylic C–H acid for Brønsted and Lewis acid catalysis
by Stephen K. Ritter
January 9, 2017
| A version of this story appeared in
Volume 95, Issue 2
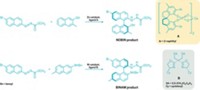
When chemists need a superstrong acid, they often reach for triflic acid, CF3SO3H; triflimide, (CF3SO2)2NH; or perhaps tris(triflyl)methane, (CF3SO2)3CH. These acids and the salts made from them are sought after as electrolytes in fuel cells, stabilizers of reactive species, and as catalysts. In seeking to diversify the strong organic acid family, researchers led by Denis Höfler and Benjamin List of the Max Planck Institute for Kohlenforschung have designed a new C–H acid: 1,1,3,3-tetratriflylpropene (Angew. Chem. Int. Ed. 2016, DOI: 10.1002/anie.201609923). Creating a new organic superacid typically starts with designing the corresponding anion, strategically locating electron-withdrawing substituents adjacent to a target C–H bond in such a way as to delocalize the negative charge to reduce its basicity. The triflyl group is often used because it’s one of the most electron-withdrawing groups known, and including more triflyl groups typically increases the acidity of C–H bonds. The researchers were looking to create a new acid with more than three electron-withdrawing groups and to keep the molecule symmetrical for distributing the negative charge. That led them to try installing four triflyl groups on propene, which they did in a two-step method starting from commercially available bis(triflyl)methane. The team found that tetratriflylpropene has the highest catalytic activity among other triflyl compounds tested for challenging Mukaiyama aldol reactions, Friedel-Crafts acylations, and other reactions.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter