Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Oxford chemist closing in on how a bird’s magnetic compass works
Peter Hore explains how he and others are tackling the magnetoreception mystery
by Mark Peplow, special to C&EN
June 13, 2017
| A version of this story appeared in
Volume 95, Issue 25
Migratory birds make epic journeys of thousands of kilometers each year, navigating with the help of Earth’s magnetic field. Yet the mechanism behind the biological compass that enables them to achieve this astonishing feat remains a mystery. Peter Hore of the University of Oxford is a leading proponent of the idea that this magnetic sense depends on chemical reactions involving excited radicals produced in a protein in the birds’ retinas. Mark Peplow spoke to him about the growing body of evidence behind the hypothesis.
How do we know that birds have a magnetic sense?
Experiments in the 1960s applied magnetic fields to migratory birds in cages. They tried to get out of the cages in different directions, depending on the direction of the field. In controlled experiments, birds can detect the axis of a magnetic field to within 5 degrees, and we suspect it would be more accurate in the wild, under natural conditions.
In 1978, Klaus Schulten (then at the Max Planck Institute for Biophysical Chemistry in Göttingen) suggested that the birds’ compass could be based on a radical pair. I thought it was an interesting curiosity but too abstract to take seriously. In 2000, though, Schulten and colleagues suggested that the radical pairs could form in a protein called cryptochrome (Biophys J., DOI: 10.1016/s0006-3495(00)76629-x), and suddenly there was a real molecule one could think about doing experiments on.
Vitals
Age: 62
Education: B.A., M.A., and D.Phil., University of Oxford
Current position: professor of chemistry, University of Oxford, 2002–present
Publications: More than 200
On bird migration: “Migration is a staggering concept, that birds will fly thousands of kilometers every year. We all marvel when we see flocks of birds flying over. People seem to be intrigued by the idea that it could be based on a chemical reaction.”
The competing hypothesis suggests that birds navigate with magnetic iron oxide particles, although we don’t really know where those particles are located. Very little evidence has emerged over the past 15 years that supports that as the basis for a compass in birds.
How would cryptochrome help sense a magnetic field?
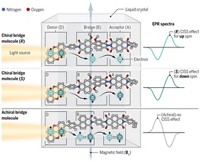
Cryptochromes are normally involved in regulating the circadian clock in birds. They contain a chromophore that absorbs blue light. In experiments on isolated proteins, blue light triggers an electron transfer within the protein to form a radical pair consisting of a flavin radical and a tryptophan radical.
The spins of the electrons in these two radicals are initially antiparallel (called a singlet state), but coupling between the electron spins and nuclear spins makes the pair oscillate between being antiparallel and parallel (called a triplet state).
The idea is that a molecule excited to the singlet state can go back to the ground state, but the triplet state cannot, so it has to react—presumably resulting in a conformational change in the cryptochrome.
The key thing is that an external magnetic field shifts the energy levels of the singlet and triplet states, and so changes the probability of relaxing back to the ground state or staying in the triplet state, and therefore affects the final outcome of the reaction. It works even though the magnetic interactions are absolutely tiny.
How would a bird perceive all this?
In plants, cryptochromes are involved in a variety of light-dependent signaling, where a conformational change affects their ability to bind to other proteins. No doubt there would be subsequent stages of biochemical signal amplification in birds after a cryptochrome binds to a receptor. But we know very little about that in the context of magnetoreception. We don’t even know what the binding partners are for the cryptochromes that might be the magnetoreceptors.
The cryptochromes must be anchored in some way, and almost certainly aligned, so that their magnetic effects reinforce rather than cancel. And for the radical pair to sense the direction of Earth’s magnetic field, which is roughly 50 microtesla, it needs to have a lifetime of about one microsecond (Proc. Natl. Acad. Sci. USA 2016, DOI: 10.1073/pnas.1600341113). I think that’s possible, in special circumstances, but I really have trouble imagining how it could be much longer than that in a biological setting.
You’ve tested the magnetic sensitivity of radical pairs in model compounds. What have you learned?
The first system we looked at where the radical pair was contained within a single molecule was based on a fullerene. Pretty much the first experiment we did showed a whopping-great magnetic field effect (C&EN, May 12, 2008, page 46), and that was really exciting. The effect depended on the direction of the field relative to the molecule, and that was crucial to show that it could be a compass rather than just a magnetic intensity sensor.
In collaboration with Leslie Dutton at the University of Pennsylvania, we recently constructed the first artificial protein-based magnetic sensor (J. Am. Chem. Soc. 2016, DOI: 10.1021/jacs.6b09682), which is much easier to manipulate than a natural protein. It has a flavin and a tryptophan at a certain distance apart, and it produces a radical pair that is magnetically sensitive, with effects similar to what we have seen in the natural protein.
So where do we go from here?
To verify this mechanism, it will have to be a multidisciplinary effort, with people from zoology all the way through quantum physics and everything in between. We need to find out which of the four cryptochromes in birds are involved in magnetic sensing, where they are in the retina, and how they are immobilized and aligned.
Experiments with transgenic flies have already demonstrated that cryptochrome is involved somehow in magnetic behaviors, so we could use RNA interference techniques to temporarily prevent cryptochromes from being expressed in a bird’s retina, and then see how that affects signaling. I think we’re within a few years of having a much clearer picture of whether this is really how it happens.
Even if it turns out that this is not the mechanism, we will have learned things about how chemical reactions respond to weak magnetic fields that I think could be applied to magnetic sensing devices based on organic materials.
Mark Peplow is a freelance writer. A version of this story first appeared in ACS Central Science: cenm.ag/hore. This interview was edited for length and clarity.







Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter