Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Cagey structures add a new dimension to silicophosphates
Adamantane-like molecule could become a favorite porous material for a range of applications
by Stephen K. Ritter
September 11, 2017
| A version of this story appeared in
Volume 95, Issue 36
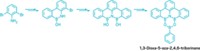
Silicophosphates are network compounds with Si–O–P linkages that form highly porous materials with enormous surface area. These materials, known as xerogels, are of interest for their possible use in making ion-conducting films for sensors, catalyst supports, biocompatible materials such as dental cement, and storage materials for confining nuclear waste. But because silicophosphates are made from moisture-sensitive starting materials and tend to be moisture sensitive themselves, their reproducible synthesis has been a challenge and only a few examples are known. Jiri Pinkas and colleagues at Masaryk University in the Czech Republic have been perfecting condensation reactions of acetoxysilanes and trimethylsilyl phosphates under anhydrous conditions to make molecular silicophosphates. In its latest work, the Pinkas group has built an adamantane-like cage molecule that has not been seen before (Inorg. Chem. 2017, DOI: 10.1021/acs.inorgchem.7b01572). When the researchers treated the silicophosphate with the Lewis acid B(C6F5)3, they found that boron coordinates to the phosphate groups to form two new compounds. One compound has three borane-phosphate pairs, but one of its P=O groups remains free and is inverted to the center of the cage, giving rise to threefold symmetry—the molecule is chiral with two enantiomers. The second compound undergoes reorganization of the Si–O–P linkages to form yet another new but smaller silicophosphate cage. The researchers anticipate that ongoing study of the synthesis, structure, and bonding of the silicophosphates will lead to additional design ideas for functional materials.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter