Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Revisiting a palladium cluster leads to an intricate surprise
Chemists find that a palladium cyclopropenium complex reported in 1964 has a more complex structure than originally thought
by Stephen K. Ritter
September 18, 2017
| A version of this story appeared in
Volume 95, Issue 37
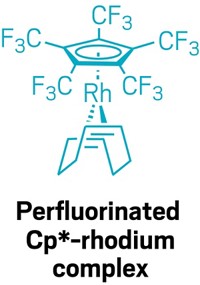
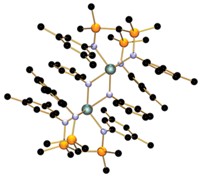
In 1964, a team of Russian chemists reported an experiment in which they treated a palladium salt with triphenylcyclopropenium chloride and ethylene. They proposed a product structure consisting of a Pd3Cl4 chain capped by two cyclopropenyl ligands. When Christian Jandl, Karl Öfele, and Alexander Pöthig of Technical University of Munich recently noticed that some reactions of cyclopropenium ions with transition metals result in ring opening and formation of four-membered metallacycles, they became interested in the actual structure of the 1964 molecule and wanted to know more about cyclopropenium’s reactivity. The Munich team repeated the originally reported synthesis and found out with the benefit of today’s analytical tools that the actual molecule is a more complex Pd6Cl8 cluster capped by triphenylpropyl ligands (Organometallics 2017, DOI: 10.1021/acs.organomet.7b00525). The researchers believe the ring-opened ligand forms in a two-step process involving the partial reduction of Pd(II) to Pd(0) by ethylene followed by oxidative addition of cyclopropenium to palladium. They devised an alternative synthesis of the cluster using Pd(II) and Pd(0) salts with triphenylcyclopropenium chloride to confirm the ring-opening event. Pöthig and coworkers say this molecule represents the first isolated organometallic palladium cluster compound, although that was not recognized in 1964, and one of the first transition-metal clusters in general—the concept of metal clusters wasn’t fully defined until 1966 by MIT’s F. Albert Cotton.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter