Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Physical Chemistry
ACS Meeting News: New catalyst details could help turn carbon dioxide into something valuable
by Mitch Jacoby
April 5, 2017

Credit: Brookhaven National Lab
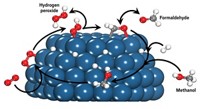
A win-win approach to curbing climate change could be capturing carbon dioxide from the atmosphere and converting it to valuable products. A study presented at the American Chemical Society national meeting in San Francisco on Monday may help advance that effort by revealing mechanistic details of a catalytic process that converts CO2 to a commodity chemical—methanol.
On industrial scales, catalysts composed of copper and zinc oxide supported on alumina hydrogenate carbon monoxide and CO2 to methanol. But these catalysts have shortcomings, according to Ping Liu, a chemist at Brookhaven National Laboratory.
Speaking at a symposium sponsored by the Division of Energy and Fuels, Liu pointed out that the Cu-ZnO catalysts are not very efficient or selective in producing methanol. These reactions also require high temperatures and high pressures of the reactant gases. What’s more, she said, chemical details of the active catalytic site remain elusive. That information could be the key to designing catalysts with improved energy and chemical efficiency, Liu says.
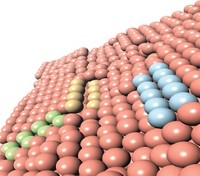
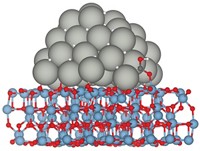
In an ongoing debate regarding the catalyst’s active site, various researchers have argued that highly active Zn-Cu alloy species are the key catalytic players. In contrast, Liu’s new work suggests that the action occurs at the atomic interface between ZnO and Cu (Science 2017, DOI: 10.1126/science.aal3573).
To reach that conclusion, Liu, Brookhaven colleagues José A. Rodriguez and Shyam Kattel, and Columbia University’s Jingguang Chen, prepared several types of Cu and ZnO reference catalysts, including one made of zinc nanoparticles deposited on copper, and another with ZnO nanoparticles on copper.
They analyzed and directly compared the CO2-to-methanol chemistry of all the catalysts using synchrotron-based photoelectron spectroscopy and computational methods. The computations predicted that Cu-ZnO surface species should be the most reactive form of the catalyst. They also predicted that the Zn-Cu species shouldn’t remain stable under reaction conditions. Instead, it should react with oxygen and form copper zinc oxide. And that’s exactly what Liu and coworkers found in the lab.
Now the group aims to use that information to optimize the interface between ZnO and Cu to improve the catalysts.
“This is a highly important study with excellent quality data and supporting theoretical calculations,” said Charles T. Campbell, a catalysis specialist at the University of Washington, Seattle. CO2 hydrogenation to methanol is one of the most likely pathways for converting the greenhouse gas to a valuable product, Campbell asserted. He added that this study should help improve that catalytic process.
Related stories:


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter