Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Molecular baskets nab nerve agent simulants
Light-initiated decarboxylation traps organophosphorus compounds in supramolecular complex
by Bethany Halford
January 1, 2018
| A version of this story appeared in
Volume 96, Issue 1
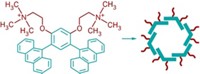
Sequestering chemical warfare agents, pesticides, and other toxic compounds could be a slam dunk, thanks to molecular baskets designed by chemists at Ohio State University. Jovica D. Badjić and coworkers created a series of basket-shaped molecules that feature a flat aromatic base fused to three bicyclic rings to form a curved unit. Three phthalimides extend this curvature into a cavitand with threefold symmetry. The phthalimides carry amino acid substituents, placing three carboxylates at the rim of each basket (shown in dotted red rings). In water, these baskets form supramolecular complexes with organophosphonates that are similar in size and shape to certain nerve agents. When exposed to light with a wavelength of 300 nm, the baskets shed the carboxylates, trapping the nerve agent simulants inside (shown, bottom). Without the carboxylates, the complexes are no longer soluble in water; they precipitate for easy removal (J. Am. Chem. Soc. 2017, DOI: 10.1021/jacs.7b11960). “In principle, one can use the concept to remove any targeted compound from aqueous solution,” Badjić says, which could have applications in controlling chemical reactivity, cleaning the environment, or even removing toxic substances from biological systems.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter