Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
As the triple quadrupole turns 40, mass spec gurus look back on what it’s meant to chemistry
Mass analyzer remains popular for quantitative applications that require sensitivity and selectivity
by Celia Henry Arnaud
March 5, 2018
| A version of this story appeared in
Volume 96, Issue 10

Christie G. Enke and Richard A. Yost didn’t set out to make a mass spectrometer. Their goal was to completely computerize an analytical instrument. It was 1975, still the early days of teaming up manually operated instruments with computers.
Enke and Yost’s report of the triple quadrupole mass spectrometer in the Journal of the American Chemical Society on March 29, 1978 (DOI: 10.1021/ja00475a072), is often credited as the birth of the instrument. But as is so often the case, the truth is more complicated.
With its sensitivity and its ability, in two stages, to select ions from a sample and then select fragments of those ions for evaluation, the triple quadrupole helped turn mass spectrometry into an analytical chemistry method. Before then, mass spectrometry had been primarily used by physical and organic chemists, not for quantifying molecules in samples but for identifying them. Over the ensuing 40 years, the triple quad became a workhorse of quantitative mass spectrometry.
Origin story
Enke, then a chemistry professor at Michigan State University, wanted to use computers to control analytical instruments. He envisioned a two-stage instrument that would separate a sample mixture in one stage and identify the components in the other.
He didn’t know what those two stages would be, but he knew he wanted to avoid chromatography.
With chromatography, he wouldn’t be able to arbitrarily select a component from the mixture and identify it. He would have to wait for it to travel slowly down and exit the chromatography column to analyze it, and if he wanted to look at another component, he’d have to do the separation all over again. “That wasn’t the kind of real-time access I was looking for,” Enke recalls.
Yost had similar ambitions, though he thought of it as “instrumentalizing” a computer. He joined Enke’s group especially to work on the computerization project. He remembered so-called quadrupole mass filters from his undergraduate advanced instrumentation class at the University of Arizona and suggested that they might work.
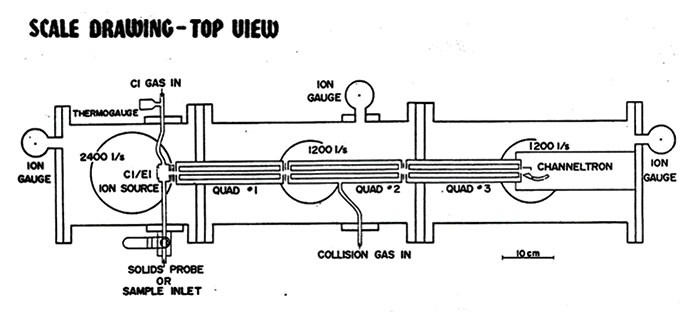
Quadrupoles are sets of four metal rods symmetrically arranged around a central axis. Each rod has a partner rod directly across from it in the configuration. Voltages applied to the pairs of rods set up an electric field that causes ions to oscillate down the central axis as they travel through the device. The applied voltages determine which ions are able to make it all the way through to the detector, effectively filtering them.
For Enke and Yost, the idea gelled during a late-night drive back from a meeting in Indianapolis. As the newest grad student in the group, Yost rode shotgun while Enke drove. They used the wee hours of the morning to talk about the project. “Somewhere between Indianapolis and East Lansing, we came up with the idea of putting two quadrupoles in a row to do ‘tandem’ mass spec,” Yost says.
That would mean selecting an ion of interest from a sample with the first quadrupole and then somehow fragmenting it and passing it to another quadrupole to identify it from its fragmentation pattern. But Enke and Yost didn’t know how to fragment the ions.
A handful of mass spectrometrists, including R. Graham Cooks of Purdue University, were already doing tandem mass spec, dubbed MS/MS, with so-called double-focusing instruments, which included an electric sector and a magnetic sector. In Cooks’s experiments, the magnetic sector selected a “parent” ion from a sample, and the electric sector analyzed fragment ions formed by collisions of those parent ions with neutral molecules such as helium. The ions had thousands of volts of kinetic energy, and each ion on average underwent only a single collision with helium, leading to low fragmentation efficiency and thus low signal in the electric sector.
Quadrupoles would produce ions with far less energy, only tens of volts, so the fragmentation efficiency would theoretically be even lower. Still, the low energy of these quadrupole ions would enable them to be controlled more easily—trapped inside a quadrupole collision chamber where they could smash into neutral molecules, as well as into the fragments being formed, multiple times. This could help boost the signal.
Enke and Yost planned to use this collision method—requiring a third quadrupole—in their instrument.
Mass spectrometrists already doing tandem MS at the time were skeptical the pair would get it working. Putting aside how to generate fragments from low-energy ions, magnetic sectors were already highly reproducible, Cooks says, whereas quadrupole mass filters had poor resolution, and the peaks in the resulting spectra shifted around and had unpredictable shapes.
That skepticism was evident when Enke and Yost tried to get funding for the project. They sent a proposal to the U.S. National Science Foundation. The eight reviews they received “were fairly consistent in their comments that this would never work,” Yost says.
But Enke and Yost succeeded in getting funding from the Office of Naval Research. ONR was less interested in the idea of a mass spectrometer than it was in the idea of computerizing an instrument.
Whatever the reasons for the funding, the duo could now proceed. “I bought a thousand pounds of stainless steel and started carving away,” Yost says.
Triple quad 101
As the names suggest, a quadrupole is a set of four metal rods, and a triple quadrupole is three sets of them in a row. The metal rods are arranged parallel to one another around a central axis.
Direct current and oscillating radio frequency voltages applied to the rods set up an electric field in the quadrupole. That field causes ions traveling through the quadrupole to follow a path that oscillates around the central axis. Ions with the right mass-to-charge ratio (m/z)—so-called resonant ions—make it all the way through a quadrupole. Others (nonresonant ions) collide with the rods. The quadrupole is tuned to different m/z values by changing the applied direct current and radio frequency voltages.
In a triple quadrupole, the first quadrupole can be used to select a particular type of ion, which is then passed into a collision cell. The collision cell can be another quadrupole, as was the case in the original design, but it no longer has to be. In the collision cell, the selected ion smashes into neutral molecules, such as helium, and forms fragments. Finally, those fragments are analyzed in the final quadrupole. Each quadrupole can be independently tuned to select ions.

Making it work
Enke and Yost’s big break resulted from a serendipitous encounter at the 1977 meeting of the American Society for Mass Spectrometry (ASMS). They ran into James Morrison, a mass spectrometrist at La Trobe University who Enke knew.

Enke and Yost described to Morrison what they wanted to do. Morrison, in return, described his experiments. Head scratching ensued over how to fragment low-energy ions.

Morrison wasn’t using quadrupoles to separate mixtures, as Enke and Yost wanted to do. He was using them to study photodissociation. In fact, he was using three of them. He used one quadrupole to select a particular type of ion, a second to confine the selected ion beam while he fragmented those ions with a laser beam, and a third to detect the desired ion fragments. But he said it was difficult to detect any fragments because the background noise was so high.
A light went on in Enke’s head. “All of a sudden, I just thought ‘My god, he’s making fragments’ ” with low-energy ions, Enke recalls. It turned out those fragments were causing the noise; they were swamping Morrison’s detector, making it difficult to distinguish the photodissociated fragments he was actually looking for.
Morrison’s noise suggested that it looked like Enke and Yost would be able to create fragments with low-energy collisions. The trick was containing the fragments in the quadrupole-based collision cell.
Because the duo’s instrument wasn’t finished yet, Yost traveled to Morrison’s lab in Australia to test whether they could actually acquire spectra from a triple quadrupole. The airfare, which ONR provided extra money for, cost the equivalent of nine months of Yost’s graduate stipend. But it paid off.
For the first set of experiments, Yost collected fragmentation spectra of cyclohexanone using Morrison’s triple quad. The instrument wasn’t set up to scan the quadrupoles over a range of mass-to-charge ratios (m/z’s)—something needed to analyze all the fragments generated from the cyclohexanone sample—so Yost had it do it by hand. It didn’t take long for him to decide to rig up a motor to do the scanning for him.
That first experiment was painful, but it worked. Enke and Yost published the data in their March 1978 JACS article.
Later that year, Yost returned triumphant to ASMS to present their work. Two audience members were Cooks and Donald F. Hunt of the University of Virginia, both of whom were consultants for Finnigan, a mass spectrometry company that became part of what is now Thermo Fisher Scientific. At a consultants’ meeting with Finnigan immediately after the ASMS conference, they encouraged the company to develop commercial versions of the instrument.
“Don Hunt wanted to build one immediately because he was interested in protein sequencing,” says Michael Story, who was head of R&D at Finnigan. “Shortly after the meeting—maybe October of that year—I took a couple of quadrupoles to Virginia and we put one together.”
When Finnigan delivered commercial triple quadrupoles, the first three instruments went to Cooks, Hunt, and Yost, who by then was on the chemistry faculty at the University of Florida. The other company with an early interest in the triple quad was Toronto-based Sciex, Enke says.
At that time, tandem MS wasn’t considered an analytical chemistry method. So it wasn’t clear how big the market for triple quad mass spectrometers was going to be. Story’s initial prediction was 10 instruments per year at a little more than $300,000 apiece. This was at a time when magnetic sector instruments could cost upward of $1 million.
What made triple quads really take off was the introduction of electrospray ionization, which could aerosolize and ionize samples coming out of a liquid chromatography instrument and pass them into a mass spectrometer. Still, electrospray didn’t fragment ions as part of the ionization process, so the triple quad’s ability to take what ions entered and fragment them was crucial for analyzing samples.
“Unless you just want your mass spec to give you molecular weights and nothing else, you really need MS/MS,” Yost says. The fragment ions generated in a triple quad can provide additional structural information about a compound. “I think that’s what really put triple quads on the map.”
Competition
The triple quad faced competition from another mass analyzer introduced commercially around the same time. The quadrupole ion trap uses electric fields to confine ions and then select particular ones to eject from the trap and send on to the detector. It also has MS/MS capabilities, but the multiple stages of ion selection happen within a single device before the ions are detected.
Finnigan was so confident about ion traps that it neglected its triple quad business and stopped further development. Although the company didn’t intentionally pull out, Story says, it ended up ceding much of the market to other companies. “We really lost our preeminent position in triple quads within five to eight years of being in the business,” he says.
“For a while, there was a race between ion traps and transmission quadrupoles,” says Terry Sheehan, a marketing manager at Agilent Technologies, a triple quad manufacturer. If Sheehan had been asked in 1995 which one would win the race, he says, “I would have told you the ion trap has the market for sure.”

Triple quads survived the competition thanks to their superiority in selectivity, sensitivity, and speed. All these advantages result from the ability to set a quadrupole to transmit only ions with a particular m/z. For quantitative assays of specific compounds, triple quads are the mass analyzer of choice. Parking the quadrupoles at electric fields that select ions with specific m/z values allows more of the ions generated to reach the detector, which improves the sensitivity.
For example, Stephen Blanksby, a mass spectrometrist at Queensland University of Technology, harnesses the triple quad’s selectivity and sensitivity for lipidomic applications. Lipids are particularly tricky to distinguish because many of them have the same elemental composition or the same nominal mass. The selectivity of triple quads gives him confidence in the molecular structure as he’s trying to identify lipids.
“We had one example a few years ago that I still get excited about,” Blanksby says. “We were able to look at the lipid profile of a single cell because we used the triple quadrupole function to remove the chemical noise and just look at the lipid signal.”
In addition to controlling the ions that go into the triple quad’s collision cell and come out of it, Blanksby uses chemical reactions in the collision cell to provide yet another layer of selection in his analysis. He uses ozone vapor in the collision cell to react with double bonds in lipids. That reaction causes a mass change that can be detected by the last quadrupole.
Triple quads are now what Story calls commodity instruments—ones that are run to answer a fixed question, such as, “Is a particular pollutant in the environment or a particular drug metabolite in this sample?” Annual sales of triple quads today are somewhere between $800 million and $1.2 billion, which translates to approximately 3,000 to 4,000 instruments per year, according to Puneet Souda, director of equity research for life science tools and diagnostics at Leerink Partners, an investment bank focused on the health care sector.
Advertisement
“You line up 50 of these instruments in the laboratory and run thousands of samples. The value of any one sample is very low,” Story says. “The current use of them will go on as long as they’re cheaper than any other way of doing it. People don’t have to revise their methods. They don’t have to recertify them to some regulatory body. They don’t have to retrain people.”
Triple quads may be commodity instruments, but even after 40 years, companies continue to improve them. At the 2017 ASMS meeting, for instance, Agilent introduced its Ultivo tandem quadrupole, which is compact enough to fit in a chromatography stack.
The development process has been made easier by improvements in theoretical modeling, says Shane Tichy, Agilent’s R&D manager for liquid chromatography/mass spectrometry. “We can build things in these programs and get a feeling for how ions behave in various regions of pressure, which then allows us to make changes to the hardware itself.”
Agilent focuses particularly on the ion guides that are used to transmit ions into the first quadrupole. “If we get high collection and high transfer efficiency, then instruments are more sensitive because we’re collecting more of the ion signal,” Tichy says.
And there’s still room to make them better. Even with improvements in ion collection and transmission, triple quad efficiency—the fraction of molecules at the front end that are analyzed as ions at the back end—is only about 10–3, Cooks says. “People still have an appetite for better sensitivity out of a mass spectrometer. Sensitivity is an efficiency characteristic,” he says. “There’s room for improvement and presumably there are people who want to do something about that.”
Enke’s original vision of a computerized instrument still hasn’t been completely realized. He wanted an instrument where an operator wouldn’t have to tell it what to do but rather what he or she wanted to know. The instrument itself, consulting a database, would come up with the sequence of operations needed to get an answer.
“We can still go a long way in that direction,” Enke says. “It’s like driverless cars. There’s more we could automate.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter