Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Looking for cheaper routes to malaria medicines
Efforts to produce low-cost synthetic artemisinin gain momentum with help from Gates Foundation
by Mark Peplow, special to C&EN
March 12, 2018
| A version of this story appeared in
Volume 96, Issue 11

In 2016, malaria killed an estimated 445,000 people, 90% of them in Africa. That’s one death every 71 seconds, caused by a disease that is both preventable and treatable. Although that death toll has fallen by 25% since 2010, the World Health Organization recently warned that progress has stalled and called for a redoubled assault on this scourge.
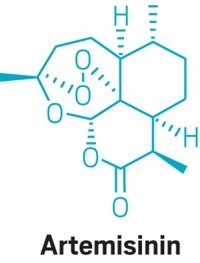
Some of the most crucial weapons in this battle are artemisinin-based combination therapies (ACTs), judged to be the most effective antimalarial medicines. They contain derivatives of artemisinin, a molecule found in the plant sweet wormwood (Artemisia annua) that is grown mostly in China and Vietnam.
But over the past 15 years, the world’s supply of artemisinin has been wildly erratic. As more farmers planted A. annua to meet rising demand, artemisinin supplies rose and pushed prices down. So farmers switched to more lucrative crops. Artemisinin supplies then plummeted and prices soared again. This roller coaster in artemisinin supplies has hampered efforts to get ACTs to everyone who needs them.
An alternative source of artemisinin could solve the problem. Back in 2004, the Bill & Melinda Gates Foundation funded a project to develop a genetically engineered yeast that could make artemisinic acid, a compound a few chemical steps away from artemisinin itself. Sanofi eventually commercialized this process to make semisynthetic artemisinin (SSA) in 2013, with the capacity to meet about one-third of global demand. Yet the anticipated ramp-up in production did not happen—in part because SSA has remained more expensive than agricultural artemisinin.
Now there is a renewed push to develop a technological solution to artemisinin production that will deliver this vital molecule at a much lower price. Last year, the Gates Foundation issued a request for proposals for novel artemisinin manufacturing technologies, offering a total of $4.5 million in grants for intensive, yearlong research programs. Some of those projects have been funded and are now under way; others are waiting for a decision in the coming weeks.
Meanwhile, researchers in Germany who have spent years developing a flow chemistry process to make artemisinin have just unveiled an upgrade to their method. They say this modified approach could dramatically boost the yield of artemisinin derived from plants.
It can’t come soon enough. Unitaid, a global health initiative, predicts that artemisinin demand will rise from 176 metric tons in 2017 to 221 metric tons in 2020, as aid agencies allocate more funding for ACTs. “There’s an ethical obligation to make this as cheaply as possible,” says Kai Rossen, editor-in-chief of Organic Process Research & Development, who previously worked for Sanofi and coauthored a paper describing the development of its SSA process (Org. Process Res. Dev. 2014, DOI: 10.1021/op4003196).
The second race for synthetic artemisinin is on.
Fresh ideas
The Gates Foundation initiative aims to improve on the current method for making SSA. The first stage of that method involves fermenting an engineered yeast, which was developed from a partnership that included the biotech company Amyris and the University of California, Berkeley. Amyris’s original proposal for the second stage—converting the yeast’s artemisinic acid into artemisinin—was deemed unsuitable for scale-up. Instead, Sanofi developed a process that begins by converting that artemisinic acid into dihydroartemisinic acid (DHAA) and finishes with an innovative photochemical reaction, generating reactive singlet oxygen to trigger a complex reaction cascade that produces artemisinin.
Sanofi scaled up this process at a facility in Garessio, Italy, making about 370 kg of artemisinin in every batch, with a yield of 55% from artemisinic acid. The price of SSA was set at a “no profit, no loss” value of $400 per kg, which partly reflected the costs of manufacturing and was carefully tailored to avoid drastically undercutting agricultural suppliers.
In 2013, SSA made up roughly 8% of global artemisinin supplies, but that fell to zero in 2015—largely because of its relatively high cost but also because other ACT manufacturers were reluctant to buy SSA from their rival, Sanofi. The following year, Sanofi sold its Garessio factory to Huvepharma, which was already contracted to carry out the yeast fermentation stage. In 2017, the facility produced around 20 metric tons of SSA, according to Nicola de Risi, CEO of Huvepharma Italia.
So the Gates Foundation has started looking for fresh ideas that could “provide a truly sustainable low cost supply of semisynthetic artemisinin,” according to its request for proposals. Representatives of the foundation declined to speak with C&EN for this story. Overall, the foundation wants a technology that can make at least 50 metric tons of artemisinin per year for $100 per kg or less, using techniques that exploit existing manufacturing infrastructure. The foundation will initially fund the projects for one year, by which time the teams must demonstrate proof-of-principle systems that are amenable to scale-up. The most successful projects would then receive further funding to continue the work.

Two biotech companies have already won grants. Manus Bio plans to engineer Escherichia coli to make artemisinic acid, offering a potentially cheaper and more efficient alternative to Amyris and UC Berkeley’s yeast. Meanwhile, Phyton Biotech has received $400,000 to find out if it can manufacture artemisinin directly by fermenting cells from A. annua itself. Plant cell fermentation is more temperamental than brewing workhorse microbes like E. coli, but Phyton has a strong track record—they are the world’s leading supplier of the cancer drug paclitaxel, made by fermenting cells from the Chinese yew tree (Taxus chinensis var. mairei).
Other groups are still waiting for a decision from the Gates Foundation. Thomas Wirth and Rudolf K. Allemann at Cardiff University hope to build on their lab-scale chemical synthesis of artemisinin. It is shorter than previous routes, thanks to a versatile enzyme called amorphadiene synthase that knits together the beginnings of the artemisinin ring system in a single step (Bioorg. Med. Chem. 2017, DOI: 10.1016/j.bmc.2017.03.068).
Rossen has advised on a different bid, led by Janine Cossy at City of Paris Industrial Physics & Chemistry Higher Educational Institution and Zacharias Amara of the National Conservatory of Arts & Crafts. Amara is keeping tight-lipped about the details until they get a verdict from the Gates Foundation, but he says that their process will convert artemisinic acid into artemisinin more efficiently than the current SSA approach.
Huvepharma Italia’s de Risi says the company has also submitted a proposal, although he too is keeping the details under wraps. But the company is in close contact with Amyris, which continues to work on artemisinin with support from the Gates Foundation. Chris Paddon, principal scientist at Amyris, says the firm is trying to reduce the cost of producing artemisinic acid by tweaking biochemical bottlenecks in its yeast, as well as improving the industrial fermentation process (Front. Plant Sci. 2018, DOI: 10.3389/fpls.2018.00087).
If any of these projects eventually deliver synthetic artemisinin at $100 per kg, it could greatly increase the availability of ACTs, says Scott Webster, an expert in supply chain logistics at Arizona State University who has modeled the effect of different interventions on the artemisinin market (Prod. Oper. Manage. 2016, DOI: 10.1111/poms.12574). “If the price gets that low, we should see a significant uptick in the use of SSA,” he says.
A plant in flow

Webster’s model of the artemisinin supply chain also sounds a note of caution for SSA enthusiasts. It should help stabilize the market, but only if it makes up a substantial share of global artemisinin production. Otherwise, SSA may not be able to offset the decrease in agricultural supply as farmers react to falling prices and exit the market.
Kerry Gilmore and Peter H. Seeberger at Max Planck Institute of Colloids & Interfaces think they can escape this catch-22 by using chemistry to boost the amount of artemisinin derived from agricultural production. Dried A. annua leaves contain about 1% artemisinin by mass, but they can also carry similar amounts of DHAA. Farmers typically dry their harvested crop in fields for about three weeks, which conveniently allows sunlight to convert some of the DHAA into extra artemisinin. But managing that process is difficult: Longer drying times can actually reduce artemisinin yields if leaves rot or blow away, for example.
Gilmore and Seeberger’s process would use industrial chemistry to replicate what the outdoor drying process achieves. In 2012, Seeberger published a photochemical process that converted DHAA into artemisinin. It was similar to Sanofi’s chemistry but operated as a flow process instead of in batches (Angew. Chem. Int. Ed. 2012, DOI: 10.1002/anie.201107446). Believing that a flow approach would significantly lower the price of artemisinin, he founded a company, ArtemiFlow, to develop the technology.
Last month, the researchers announced that they can now feed crude extracts of A. annua into their photochemical flow reactor and rely on chlorophyll compounds from the plant itself to serve as photosensitizers that catalyze the reaction. The reaction sequence takes just 15 minutes and offers artemisinin yields of around 65% (Angew. Chem.Int. Ed.2018, DOI: 10.1002/anie.201801424). “I thought it was fascinating to do it like that, effectively putting the plant into a flow process,” Rossen says. “But there are still many challenges to address before this could be commercialized.”

If the process can improve artemisinin yields per hectare, however, it may provide a way to offset any fall in artemisinin price and give farmers a stronger economic incentive to keep growing A. annua. “I think it has the potential to be quite powerful,” Arizona State’s Webster says. Moreover, chlorophyll is such a potent photosensitizer that it could catalyze the conversion of far more DHAA than is present in plant extracts. In principle, the crude feedstock from the leaves could be supplemented with DHAA derived from the output of bioengineered yeast, Gilmore says, making the process more economical.

ArtemiFlow now hopes to commercialize this process in collaboration with the Kentucky Tobacco Research & Development Center, which is looking for new crop opportunities for local farmers. The center will run field trials of several A. annua strains this year. “We have to assess the economics of production, see what the yields will be here in Kentucky,” says Orlando Chambers, the center’s executive director. Although ArtemiFlow did not bid for the Gates Foundation grants advertised last year, the company hopes to attract separate funding from the foundation to help pilot and scale up its process.
Solving the artemisinin supply problem remains a crucial part of tackling malaria, and Gilmore is confident the approach can deliver. “We will commercialize this on the 50-ton scale,” he vows. “It’s viable, it’s efficient, and it’s scalable.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter