Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Study elucidates mechanism of MOF-mediated catalysis
Findings could lead to custom-designed catalysts with improved activity and selectivity
by Mitch Jacoby
March 19, 2018
| A version of this story appeared in
Volume 96, Issue 12
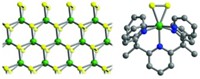
In addition to garnering widespread attention as materials for gas storage and separation, metal-organic frameworks (MOFs) have shown themselves to be worthy catalysts in a limited number of reactions. MOFs’ catalytic usefulness could be boosted by design, if researchers understood how these crystalline porous materials drive reactions. But many details remain unknown. So a team led by Christopher J. Cramer and Laura Gagliardi of the University of Minnesota, Twin Cities, and Bruce C. Gates of the University of California, Davis, coupled spectroscopy and computational methods to ferret out mechanistic details of a test reaction—ethanol dehydration on the MOFs UiO-66 and UiO-67 (J. Am. Chem. Soc. 2018, DOI: 10.1021/jacs.7b13330). These MOFs contain Zr6O8 clusters joined by dicarboxylic acid linkers. Because MOF clusters function as catalysts when they have defects—for example, vacancies due to missing linkers—the researchers deliberately introduced vacancies synthetically. Analysis of the resulting MOFs shows that they dehydrate ethanol selectively, forming diethyl ether, not ethylene, the competing product. The team notes that the key to selectivity is having adjacent vacancies, which enables ethanol molecules to bind to neighboring sites on the clusters and facilitates ether formation via an SN2 mechanism.
CORRECTION: This story was updated March 20, 2018, to correct Bruce C. Gates’s affiliation. He is a professor at the University of California, Davis, not the University of California, Irvine.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter