Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Graphite-bound molecules combine best of homo- and heterogeneous catalysis
Conjugated pyrazine linkage endows tunable molecular system with metal-like electron-transfer properties
by Mitch Jacoby
January 29, 2018
| A version of this story appeared in
Volume 96, Issue 5
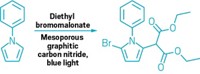
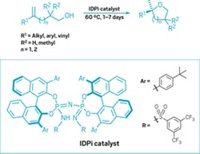
Most synthetic catalysts fall squarely into one of two categories—homogeneous or heterogeneous, each with its characteristic strong points. Few catalytic systems capitalize on the best of both worlds. Now, a study led by chemists at Massachusetts Institute of Technology takes a key step in that direction (J. Am. Chem. Soc. 2017, DOI: 10.1021/jacs.7b10723). Heterogeneous catalysts, especially ones used in electrochemistry, tend to be robust metals that efficiently bind reactant molecules and mediate catalytic reactions via electron transfer. But molecular details of the binding and active sites are tough to pin down and their structures often change during reaction. In contrast, the structures of metallo-organic molecules that serve as homogeneous solution-phase catalysts are well studied and can be modified easily via synthesis to tune catalyst performance. When used as electrocatalysts, however, electron transfer from the electrode through the catalyst to the reagent molecule is a stepwise, thermodynamically unfavorable process. To bypass that problem, a team led by MIT’s Yogesh Surendranath condenses o-phenylenediamine derivatives with o-quinone moieties commonly found on graphitic materials. The procedure attaches catalytic molecules to graphite electrodes via strong conjugated pyrazine linkages. The group’s graphite-bound catalysts, which have been used to mediate fuel-cell reactions and convert CO2 to CO, exhibit electron-transfer properties identical to those of heterogeneous metal catalysts but retain the tunability of homogeneous catalysts.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter