Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biochemistry
New mechanism proposed for old diabetes drug
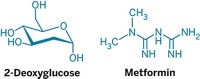
Type 2 diabetes drug metformin impedes glucose overproduction in a redox-dependent manner, researchers say
by Cici Zhang
July 27, 2018
| A version of this story appeared in
Volume 96, Issue 31
Scientists have proposed many mechanisms of action to explain the glucose-lowering benefits of metformin, the first-line treatment for type 2 diabetes. But previous studies typically used doses much greater than those given therapeutically. Now, studying rats receiving clinically relevant doses of metformin, researchers report the compound blocks conversion of select substrates into glucose in a redox-dependent manner. This would not be consistent with other proposed mechanisms by which metformin inhibits gluconeogenesis, a process that turns noncarbohydrate carbon substrates into glucose in the liver and is abnormal in type 2 diabetes, the researchers say (Nat. Med. 2018, DOI: 10.1038/s41591-018-0125-4). Gerald Shulman of Yale University School of Medicine and colleagues tracked 13C-labeled substrates in both diabetic and nondiabetic rodents. They found metformin inhibited liver gluconeogenesis starting from a subset of substrates whose conversion to glucose relies on the redox state of liver cells’ cytosol. They also showed that metformin treatment made the cytosol more reduced. When the team prevented this more-reduced state of the cytosol, metformin no longer could block liver gluconeogenesis. Shulman says these results suggest that new therapies targeting the liver’s cytosolic redox state might be an effective way to treat type 2 diabetes.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter