Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biotechnology
Microfluidic device brews human embryo-like structures
The cell culture system provides scientists with a way to model the earliest stages of human embryo development without stepping over ethical boundaries
by Alla Katsnelson
September 13, 2019

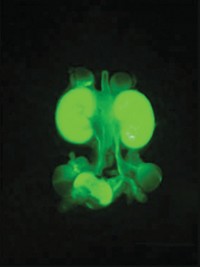
Researchers have created a microfluidic device that can reliably make cell structures that are similar to early-stage human embryos without using actual embryonic tissue (Nature 2019, DOI: 10.1038/s41586-019-1535-2).
In the five days after a human egg cell is fertilized, it divides repeatedly into hundreds of cells to form a hollow structure called a blastocyst. Once a human blastocyst implants into a uterus a few days after that, it essentially goes dark to science, because there is no way to visualize it. Ethical guidelines prohibit growing human embryos for longer than 14 days in the lab, so studying this process in a dish has also been a nonstarter.
The limited ability to study embryos “has really prevented us from learning a lot about early human development,” says Jianping Fu, a bioengineer at the University of Michigan, Ann Arbor, who led the new study. “And unfortunately, during this time, many miscarriages can happen and serious birth defects can form.”
The new microfluidic device may help shed light on these early steps in human embryo development. The device exposes stem cells derived from human skin to a chemical signal, triggering the cells to self-organize into structures that develop hallmark features of human embryos in the first few weeks after fertilization.

In earlier work, Fu and his colleagues grew so-called induced pluripotent stem cells—cells that they had transformed into stem cells from another form such as skin cells—in a 3-D cell culture system. They were surprised to find that about 5% of these cells spontaneously formed asymmetrical structures that resembled early embryos. Fu, an expert in designing microfluidic systems, set out to build a device that produces these structures in a more reproducible and controlled way.
As microfluidic devices go, what the team built is quite simple, Fu says. It has three channels: one into which the scientists load the pluripotent cells, one that holds the chemical signals, and one in which the cells get exposed to the signals. “We can control when and where cells in the colony are stimulated by chemical signals,” Fu explains.
Researchers previously determined that a group of pluripotent stem cells can self-organize into a cell structure mimicking the epiblast, which forms within the blastocyst and has the potential to generate the different cell types of the embryo. Additional signals, either chemical or mechanical, can then trigger some of the cells to push the whole group down a developmental path mimicking that of a primitive human embryo.
The signal Fu and his colleagues used was a molecule called bone morphogenetic protein 4, which they identified in previous studies. It initiates the differentiation of cells destined to form the amniotic tissue, Fu explains.
The fact that the process works on pluripotent cells derived from skin cells allows researchers to study early embryo development without using tissue taken from actual embryos. The cell structures formed in the microfluidic device also lack certain cell types needed to form viable embryos, he notes. As a result, the microfluidic system avoids any ethical issues and can recapitulate embryo development 1 to 3 weeks after fertilization.
“The concept of being able to model some of these early events of human development is really quite exciting,” says Janet Rossant, a developmental biologist at the Hospital for Sick Children in Toronto, who was not involved in the research. “Other people have been able to make such 3-D models, but not in such controlled manner.”
In addition to shining a light on the basic biology of early human development, the system is a much-needed tool for studying glitches that arise in early pregnancy, such as miscarriages. It could also be used to test whether drugs are safe for use in early pregnancy, Fu says.
CORRECTION
This story was updated on Sep. 19, 2019, to correct the name of the chemical signal used in the microfluidic device. It is called bone morphogenetic protein 4.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter