Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Epigenetics
Human epigenetics may play role in MRSA infection outcomes
DNA-methylation signatures can distinguish between persistent and resolving MRSA infections
by Celia Henry Arnaud
March 4, 2021
| A version of this story appeared in
Volume 99, Issue 8

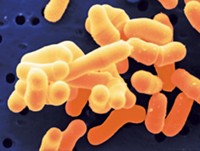
It’s not clear what factors cause one infection with methicillin-resistant Staphylococcus aureus (MRSA) to linger and another to resolve. A new study suggests that infected people’s DNA methylation patterns, which modulate gene expression, may play a role. Elaine F. Reed of the University of California, Los Angeles, Michael R. Yeaman of the Lundquist Institute for Biomedical Innovation at Harbor-UCLA Medical Center, and coworkers compared the methylation pattern of DNA in circulating immune cells from people with either persistent or resolving MRSA infections (Proc. Natl. Acad. Sci. U.S.A. 2021, DOI: 10.1073/pnas.2000663118). The researchers found 276 sites in the DNA that were methylated to different extents in the two patient populations. Of these sites, 16 were near transcription factor binding sites associated with modulating immune function, with different transcription factors involved in persistent and resolving infections. It’s not yet clear how the sites affect the response to MRSA infections, but for people with persistent infections, the team found reduced methylation at sites that have previously been shown to suppress protective immune responses, and for people with infections that resolved, at sites that are known to be involved in the stress response to sepsis. The researchers don’t know if the methylation signatures preceded or were caused by the infections or whether the signatures depend on the specific antibiotic treatment used. Nonetheless, the findings are an important step toward enabling personalized antibiotic therapy.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter