Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Infectious disease
Covid-19
What explains the non-respiratory symptoms seen in some COVID-19 patients?
Some patients experience neurological, cardiovascular, and intestinal symptoms after infection with SARS-CoV-2
by Megha Satyanarayana
April 14, 2020
While COVID-19, the disease caused by the novel coronavirus SARS-CoV-2, is generally a respiratory infection with coughing and shortness of breath as key features of the illness, some people have reported other symptoms, including loss of smell, heart trouble, and diarrhea.
These less common symptoms, especially cardiac ones, have caused hospitals to adapt their intake protocols to spot possible COVID-19 patients, says Salim Virani, a cardiologist at Baylor College of Medicine. “Almost all hospitals that I’ve talked to, everybody has a protocol whereby they are testing these patients for COVID-19 as well,” Virani says about people who arrive at the emergency department with heart concerns.
Scientists say that these non-lung symptoms might also be good indicators of SARS-CoV-2. Once inside a person’s body, the novel coronavirus attaches to a protein on human cells called angiotensin-converting enzyme 2 (ACE2). This enzyme, which is normally involved in blood pressure regulation, sits on the surface of different types of cells, including those in the brain, blood vessels, heart, intestines, and kidneys.
It’s plausible that the novel coronavirus causes these other symptoms by directly infecting some of these cells. But this infection also can cause a massive immune response, leading to the overproduction of small immune molecules called cytokines. This cytokine storm triggers inflammation and organ damage. It’s been observed in the lungs of the sickest COVID-19 patients, but it likely happens in other organs as well.
“If you have a severe infection, and there is a systemic response related to that, inflammation can be protective early on,” Virani says. “But then that response itself, when it’s a systemic inflammation all over the body, can become detrimental.”
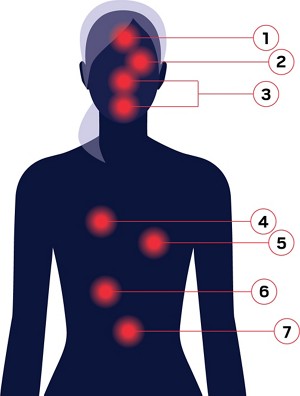
Here is a look at some of the body parts where the novel coronavirus might cause other symptoms in people, and what scientists think is behind these symptoms. Because researchers are still learning about SARS-CoV-2, a lot of what they hypothesize is based on previous characterizations of the virus’s cousins, the virus that caused the severe acute respiratory syndrome (SARS) outbreak from 2002 to 2004, and the coronavirus that causes Middle East respiratory syndrome (MERS).
Support nonprofit science journalism
C&EN has made this story and all of its coverage of the coronavirus epidemic freely available during the outbreak to keep the public informed. To support us:
Donate Join Subscribe
Central nervous system
Some COVID-19 patients have shown up at the hospital confused, or with what doctors call an altered mental status. For example, a flight attendant in her fifties from Detroit developed something called acute hemorrhagic necrotizing encephalopathy. This brain condition is characterized by inflammation and tissue damage and is often linked to viral infections. Brent Griffith, a neuroradiologist at Henry Ford Hospital, led the team of health care workers who described the woman’s condition, which they attribute to cytokine storm and a breakdown of the blood-brain barrier. Using magnetic resonance imaging, they showed damage to her brain, and noted that while they were unable to test for the presence of SARS-CoV-2 in fluid from her central nervous system, the presence of virus isn’t always necessary for acute hemorrhagic necrotizing encephalopathy (Radiol. 2020, DOI: 10.1148/radiol.2020201187).
Another research team, led by Yan-Chao Li of Jilin University, thinks that based on what we know about where in the body the virus behind the SARS outbreak attacks, SARS-CoV-2 might be able to infect the central nervous system. Li and the team also propose that when the coronavirus infects the brain, it may damage the nerves that regulate breathing, and that this might be one of the reasons why some people end up in respiratory failure (J. Med. Virol. 2020, DOI: 10.1002/jmv.25728).
Eyes
There have been some reports of eye inflammation, or conjunctivitis, associated with COVID-19, but these symptoms have been rare. Still, some researchers have wondered whether the virus could get into the body through the eyes. In small studies in China, researchers have detected the virus’s RNA in people’s tears (JAMA Ophthalmol. 2020, DOI: 10.1001/jamaophthalmol.2020.1291).
Eye cells do express the ACE2 receptor, but these scientists still think eye-related infection or tear-related spread of the virus would be quite rare. Led by Chuan-bin Sun of the Zhejiang University School of Medicine, this team suggests that tears would wash away the virus, and that immune mechanisms in the eyes, led by antibodies and a molecule called lactoferrin, would prevent widespread infection (J. Med. Virol. 2020, DOI: 10.1002/jmv.25859). The American Academy of Ophthalmology has also weighed in on the issue, saying that, “infectious virus has not yet been cultured from the conjunctiva of any COVID-19 patient.”
Taste and smell
Danielle Reed of the Monell Chemical Senses Center says that viral infections often affect our sense of smell, and thus taste, because the infection blocks airways, preventing odorants from interacting with smell receptors. However, in the case of COVID-19, she says, researchers haven’t ruled out direct infection of smell-sensing cells, or some other mechanism affecting the nerves that conduct smell signals to the brain.
So, Monell, as part of the Global Consortium for Chemosensory Researchers, is gathering data from people who have tested positive for COVID-19, and who are experiencing loss of smell and taste.
While olfactory neurons do not express ACE2, cells that surround those neurons, and provide structural support, do. The virus could infect and kill these sustentacular cells, weakening the scaffolding that holds olfactory neurons up, and perhaps affecting the neurons’ function. “We envision that they’re taking up the virus,” Reed says. “But, how it’s causing problems with people’s ability to smell is not understood.”
There is precedence for these sensory symptoms, Reed points out. People with SARS also reported a loss of smell.
Cardiovascular system
Some people with COVID-19 have had heart attacks, but Baylor’s Virani says that early reports of patients in Seattle and New York indicate that arrhythmia, an irregular heart beat, is the most common cardiovascular symptom. Doctors have also observed inflammation of heart tissue, or myocarditis.
Heart muscle cells express ACE2, Virani says, and it is possible that SARS-CoV-2 is infecting the heart, but he can’t rule out cytokine storm and what’s happening in the lungs as the culprits behind these cardiac symptoms. “The severity of the heart muscle involvement, in most cases, but not in every case, is actually directly related to the severity of the lung involvement,” he says, adding that in some patients, as their lung disease progresses, markers for cardiac damage also increase. Doctors are treating some of the people with these cardiac symptoms with drugs that block some of the perpetrators of cytokine storm, including interleukin-6.
Liver
In one study of 99 people infected with SARS-CoV-2 in Wuhan, 43 had elevated levels of the liver enzymes alanine aminotransferase, aspartate aminotransferase, and lactic dehydrogenase (Lancet 2020, DOI: 10.1016/S0140-6736(20)30211-7). High levels of these enzymes are a sign of liver damage. In another study of nearly 1,100 people who tested positive for the novel coronavirus, between 28% and 40% had elevated liver enzymes, and the more critically ill they were, the more likely they were to have higher levels (N. Eng. J. Med. 2020, DOI: 10.1056/NEJMoa2002032 .
Jie Li, who is an infectious disease scientist at Shandong University, and Jian-Gao Fan of the Shanghai Jiao Tong University School of Medicine, wrote in a recent paper that there aren’t many cells in the liver that express ACE2, so it’s not clear if the virus is causing the damage directly itself (J. Clin. Transl. Hepatol. 2020, DOI: 10.14218/JCTH.2020.00019). They point out that cytokine storm might affect the liver, as could sepsis, which is a large scale bacterial infection that seems to trail coronavirus infection in some people.
Some people who have had SARS or MERS also experienced liver damage, but it’s still too early to say what’s happening with SARS-CoV-2 and the liver.
Gastrointestinal system
As the COVID-2 outbreak spread through Hubei province in China, a team of researchers, led by Lei Tu of Huazhong University of Science and Technology, surveyed about 200 people with COVID-19 to see if they experienced gastrointestinal symptoms and found that 19% reported some combination of diarrhea, vomiting, or abdominal pain. That data makes gastrointestinal distress perhaps the most common non-respiratory symptom of SARS-CoV-2 infection. Their analysis also indicated that some patients went to the hospital complaining about these gastrointestinal issues and no respiratory symptoms, suggesting that some people with COVID-19 could have been missed by doctors. In fact, six of the people they studied never had any respiratory symptoms.
Tu and colleagues found viral RNA in about half of the stool samples they tested in their study. The team speculates that the inflammatory response to infection could explain intestinal symptoms, as the lungs and gut are linked via the body’s mucosal system in what researchers call the gut-lung axis. But scientists also think it’s possible that the novel coronavirus infects intestinal cells carrying the ACE2 receptor. One study of human tissue from COVID-19 patients found evidence of novel coronavirus proteins inside gastrointestinal cells that express ACE2, suggesting that the virus can infect the human gut. That team concluded that these data suggest gastrointestinal symptoms should not be overlooked in the absence of respiratory illness (J. Gastro. 2020, DOI: 10.1053/j.gastro.2020.02.055).


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter