Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Microbiome
Serotonin helps gut microbes thrive
The antidepressant Prozac may interrupt this gut-microbe communication
by Megha Satyanarayana
September 4, 2019
| A version of this story appeared in
Volume 97, Issue 35
From the crowded confines of the intestine comes new research about a bacterial species that uses serotonin from the gut to gain an upper hand in its quest to colonize and survive.
In studies done in mice, a team of researchers found that Turicibacter sanguinis, a common gut microbe, can signal nearby intestinal cells to release serotonin, a neurotransmitter typically associated with mammalian mood and digestion (Nat. Microbiol. 2019, DOI: 10.1038/s41564-019-0540-4). When the bacteria take up serotonin, says Elaine Hsiao, the lead researcher on the project at the University of California, Los Angeles, the small molecule prompts the expression of genes related to growth and survival, allowing the microbe to colonize its host’s gut.
Treating the mice with the popular antidepression drug, fluoxetine, sold as Prozac, blocks this uptake, and prevents T. sanguinisfrom colonizing the animals. The Prozac findings may help explain some of the drug’s gastrointestinal side effects, Hsiao says.
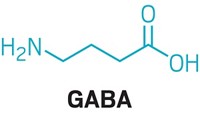
Serotonin is not the first mammalian neurotransmitter that has been discovered to broker a relationship between gut microbes and their hosts, Hsiao says, but this serotonin communication is critical. Up to 90% of the body’s serotonin comes from gut cells, and a metabolite from T. sanguinis regulates about 50% of that production. It’s an example of co-evolution between microbes and their host in which the microbe can use something produced by the host to outcompete other species, she says.
“It’s always a fitness game—who gets to be more of the population. The gut microbes that promote serotonin are the ones that benefit from it,” she says.
Hsiao’s team did several tests to figure out how T. sanguinis uses serotonin. Mice, human, and T. sanguinis cells all have proteins in their membranes that transport serotonin from outside of the cell to inside of it. Mice missing their version of this serotonin transporter had high levels of serotonin in their guts, leading to high levels of bacteria like T. sanguinis. Adding that transporter to bacteria that normally don’t carry it gave those microbes some ability to take up serotonin, demonstrating that the protein was the reason the neurotransmitter could get into bacterial cells. And when T. sanguinis was the only microbe colonizing the guts of germ-free mice, serotonin didn’t seem to have much effect on the bacteria, suggesting that T. sanguinis needs the neurotransmitter only when it is fighting other microbes for space.
This serotonin-mediated communication seems to have some effect on mice’s ability to metabolize lipids. Mice colonized solely with T. sanguinis had lower triglyceride levels and T. sanguinis-onlyfemale mice had smaller fat cells, compared with controls. Prozac is part of a class of drugs that blocks serotonin transporter proteins. One of the side effects of these selective serotonin reuptake inhibitors is weight gain. Hsiao wonders if these drugs may block the transporters in T. sanguinis, disrupting the communication between the microbes and the gut and, as a result, affecting lipid metabolism.
Her team is now looking at what serotonin does once it gets inside the bacterial cell and how that leads to changes in gene expression.
Philip Strandwitz, a gut microbe expert at Holobiome, a biotechnology company working on nervous system therapies involving the microbiome, says the paper is helping elucidate important steps in the communication between the intestinal nervous system and the bacteria living around it. Hsiao is an advisor to Holobiome, but Strandwitz and the company were not involved in the work.
A lot of microbiome work involves seeing some effect of a microbe on health and disease, but not really understanding the underlying mechanisms, Strandwitz says. “We might be able to put poop into something and make it dance, but we don’t know why it’s dancing, and what is the chemical process—why microbes are making that thing dance.” This study, he says, is helping researchers understand those mechanism questions.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter