Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Solar Power
Polymer solar cell hopes to claim new record
Engineering two organic molecules for better compatibility boosts the conversion efficiency of polymer solar cells to 14.2%
by Katherine Bourzac, special to C&EN
May 23, 2018
| A version of this story appeared in
Volume 96, Issue 22
By carefully tailoring the electronic properties and side-chain structure of a polymer, researchers have made one of the best performing organic solar cells yet. In tests in the lab, the polymer solar cell converts 14.2% of the energy in sunlight into an electrical current. If certified by one of the gold standard agencies for accrediting solar cell performance, such as the National Renewable Energy Laboratory (NREL), this would be a record for organic cells—but it’s not yet stable enough to take the laurels (J. Am. Chem. Soc. 2018, DOI: 10.1021/jacs.8b02695).

Organic solar cells can be transparent, lightweight, and flexible. The advantage of these materials is that many of them can be mixed with a solvent and printed on large rolls of plastic at high speed—analogous to printing newspapers. “This makes it a cheap process,” says Harald W. Ade, a physicist at North Carolina State University. But polymer solar cells haven’t been widely adopted because their efficiency remains low.
Ade is collaborating with Jianhui Hou, a chemist at the Chinese Academy of Sciences, to make more efficient polymer solar cells. These kinds of cells require two well-matched organic molecules. The first, usually a polymer, absorbs light and produces excited charges. The second, usually a small molecule, pulls electrons from the first, allowing them to flow through electrodes so the cell can generate current. The challenge is that when chemists improve, say, the light absorption of the donor, its electronic properties may no longer pair well with the acceptor, hampering the electron transfer from one molecule to the other. In other cases, the two molecules may no longer sit close enough to interact. Making a good polymer solar cell is like a whack-a-mole game: Chemical engineers solve one problem only to have another pop up.
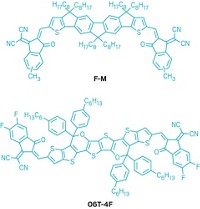
Recent progress in designing new acceptor molecules that are more efficient than the usual fullerenes has led to considerable improvements in cells’ performance. Earlier this year, Ade and Hou collaborated on a new acceptor molecule called IT-4F whose fluorine groups created a strong pull on electrons, encouraging them to hop away from the donor and boosting current (Adv. Mat. 2018, DOI: 10.1002/adma.201707170). But they still had a problem: when paired with a conventional donor polymer, the two molecules’ energy levels weren’t well matched, so the cell’s efficiency was limited.
To address this, Hou tweaked the donor polymer, based on benzodithiophene, to be a better partner for IT-4F. By adding functional groups containing fluorine, he changed the energy of electrons that the polymer could donate, enabling electrons to hop more easily from the donor to the acceptor. “This is all done by chemical intuition,” says Ade.
Next, they wanted to make sure the two molecules would align physically so they could transfer electrons in solution. Ade’s group, which specializes in studying the morphology and self-assembly of polymer materials, helped with this part of the design. They tried adding three different side chains to the polymer and settled on the 10-carbon, n-decyl chain that encouraged the molecules to form a compact structure, which improved their interaction with the acceptor.
The solar cells cast from the mixture had a conversion efficiency of 14.2% in the lab. The current official record-holder, an organic solar cell made by Henry Yan at Hong Kong University of Science and Technology in collaboration with Ade, was certified at 11.5% by NREL. Ade says his team has not sent their cells to NREL for certification because they’re not stable enough: IT-4F diffuses easily and moves around in the cell, so researchers have yet another molecular mole to whack. But Ade says this is a common problem with organic cells, and can be solved by making the small molecule bigger.
Advertisement
Yan says Ade and Hou are in good company—many labs have recently published papers describing efficiency gains in organic solar cells that use non-fullerene acceptors. None of them have shown up on NREL’s chart of champion cells but Yan expects they will be once the stability problems can be addressed. The non-fullerene designs could pave the way towards widespread commercial adoption of organic solar cells, says Yan. Although a better understanding of the materials’ chemistry and their stability problems are needed, these types of organic solar cells “are the future, that’s for sure,” he says.
“I think 18 or 20% efficiency in a couple years is in the cards—beyond that it gets very difficult,” says Ade. “The problem in this field is it’s very much based on trial and error,” he adds. Ade thinks developing better models of the self-assembly of organic solar materials will help the field advance more rapidly.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter