Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
2-D Materials
Experiments reveal how hydrogen sticks to graphene
Energy-dissipating ripple may explain how C–H bonds form on the 2-D material
by Sam Lemonick
April 25, 2019
| A version of this story appeared in
Volume 97, Issue 17
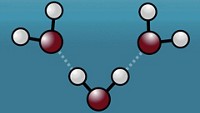
The two-dimensional sheet of sp2-hybridized carbon atoms in graphene helps grant the material unique and useful chemical, material, and electronic properties. Adding hydrogen atoms to the carbons can improve those properties, for example by turning graphene into a semiconductor.
New experimental and computational experiments reveal how energy dissipates through a graphene sheet when hydrogen atoms strike it, making C–H bond formation possible (Science 2019, DOI: 10.1126/science.aaw6378).
Alec. M. Wodtke of the University of Göttingen and the Max Planck Institute for Biophysical Chemistry was experimenting with hydrogen scattering from metal surfaces when his group observed that graphene grown on platinum behaved differently. Hydrogen and metal atoms collide like billiard balls, he says, but when hydrogen hits a graphene carbon it doesn’t bounce off, it forms a bond.
Wodtke’s collaborator Thomas F. Miller III of the California Institute of Technology used computational models to better understand what happens. When struck by a hydrogen atom, graphene dissipates energy in two ways: an up and down vibration of the sheet and a shock wave moving out across its surface from the point of impact.
The latter, Wodtke explains, is the response of neighboring carbons as the impacted carbon changes its electronic structure from sp2- to sp3-hybridization as it forms or attempts to form a bond with hydrogen. The researchers say the ripple, which only lasts about 10 femtoseconds, explains why hydrogen can stick even when it strikes the sheet at high energy, near 2 eV. Previous models predicted lower sticking probabilities. When the hydrogen does stick, according to Wodtke, it’s because the rehybridized carbon chases down the ricocheting hydrogen from behind and catches it.
This study is the first in which hydrogen “sticking probabilities can be measured for specific energies and incidence angles, which makes it a big deal,” says Bret Jackson, a chemist at the University of Massachusetts Amherst who has modeled this graphene system. Wodtke says this work is one step on a long path to better understanding how hydrogen interacts with graphene.





Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter