Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Inorganic Chemistry
Einsteinium coordination complex isolated for the first time
Experiments using scant nanograms of element 99 lead to first measurement of an einsteinium bond length
by Mark Peplow, special to C&EN
February 6, 2021
| A version of this story appeared in
Volume 99, Issue 5
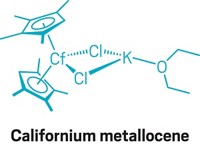
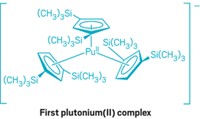
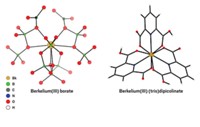
Einsteinium is the heaviest element in the periodic table that can be made in sufficient quantities for conventional chemistry experiments. Only a handful of compounds of this incredibly rare element have ever been isolated, and they’ve all been simple inorganics such as oxides and halides. But after 6 years of planning, researchers have finally created an einsteinium coordination complex (Nature 2021, DOI: 10.1038/s41586-020-03179-3). “I don’t believe we’ve seen any study on an isolated molecular complex” of einsteinium before, says Rebecca J. Abergel of Lawrence Berkeley National Laboratory and the University of California, Berkeley, who co-led the research with Stosh A. Kozimor of Los Alamos National Laboratory. The researchers worked with less than 200 ng of 254Es, which was produced at Oak Ridge National Laboratory’s High Flux Isotope Reactor by firing neutrons into a curium target. This relatively stable einsteinium isotope has a half-life of 276 days. The team treated 254Es with a hydroxypyridinone-based ligand to form an eight-coordinate complex and then carried out luminescence spectroscopy and X-ray absorption spectroscopy experiments. The scientists measured the first bond length involving einsteinium and confirmed that the complex was in the +3 oxidation state. The research helps fill data gaps in the actinide series of elements and should enable researchers to better understand chemical and physical trends across the series.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter