Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Nanomaterials
Nanoscale chains forged from a simple molecule
Assembly process can link up to 22 rings using noncovalent interactions
by Bethany Halford
July 15, 2020
| A version of this story appeared in
Volume 98, Issue 28

In an Olympic-level feat of chemical choreography, scientists have assembled nanoscale chains from a simple monomer held together only by noncovalent interactions. Making such a complex supramolecular structure from such a simple building block takes self-assembly to a new level, experts say.
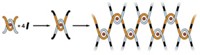
Shiki Yagai, a chemist at Chiba University who led the research effort, says the nanoscale chains, though intricately forged, are the result of an accidental discovery. “We were originally investigating methods to efficiently synthesize ring-shaped molecular assemblies,” Yagai says. A simple monomer Yagai’s team created can assemble via hydrogen bonding to make a six-membered supermacrocycle. These snowflake-like structures then stack one on top of the other, curving as they do so to make snakelike structures or helical coils. Under the right conditions, the snowflake stacks will curve into rings.
To their surprise, Yagai and coworkers observed that when they mixed certain polar and nonpolar solvents, the rings would link together. When they investigated the phenomenon, they found that the inside of the ring seeds the formation of another ring around itself. By sequentially adding more monomer to a solution of rings, they were able to drive the formation of long supramolecular chains of up to 22 links (Nature 2020, DOI: 10.1038/s41586-020-2445-z).

David A. Leigh, an expert in complex molecular topology at the University of Manchester, says it’s remarkable to see a process that threads and cyclizes such large, regularly sized rings. “It opens the door for future exploration of the properties mechanically interlocked molecular structures can have at these larger length scales,” he says.
“This is a remarkable example of topologically complex supramolecular materials,” adds M. Reza Ghadiri, who studies self-assembly at Scripps Research, California. “Given better understanding of the basic principles governing such topological self-assembly, a new generation of materials could be rationally fabricated and studied for their potentially unique properties.”
Yagai first realized the power this technique when he saw that it could make a chain with five links that resembled the symbol for the Olympic Games. “I seriously thought that this result should be published in the year of the Tokyo Olympics,” he says. “Unfortunately, the Olympics have been postponed, but I am very happy to leave a trace in 2020.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter