Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Polymers
Red blood cells catalyze polymerization
Iron from hemoglobin initiates radical reaction
by Celia Henry Arnaud
June 29, 2018
| A version of this story appeared in
Volume 96, Issue 27

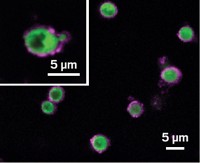
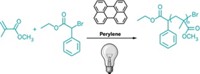
Performing polymer synthesis in biological systems could provide a biocompatible approach for engineering cells for therapeutic or other applications. A team led by Greg G. Qiao of the University of Melbourne has used components from blood to catalyze the synthesis of poly(acrylates) and poly(acrylamides) via reversible addition-fragmentation chain transfer, or RAFT, radical polymerization (Angew. Chem. Int. Ed. 2018, DOI: 10.1002/anie.201802544). The researchers chose RAFT polymerization because it works in water-based systems with many types of monomers. To kick off the in vitro reaction, they add the enzyme glucose oxidase to produce hydrogen peroxide. The team proposes that the H2O2 degrades the heme group in hemoglobin, releasing iron ions that eventually produce hydroxyl radicals, the initiators for the polymerization, via a Fenton reaction. Purified hemoglobin, isolated red blood cells, and whole blood all catalyzed polymer formation. The researchers plan to use the reaction for encapsulating biomolecules and cells. Craig Hawker, a polymer chemist at the University of California, Santa Barbara, says the method “opens up the very exciting possibility of using hemoglobin in combination with endogenous reactive oxygen species to productively hijack biological processes for in vivo polymer synthesis.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter