Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Drug Discovery
New class of synthetic antibiotics battle drug-resistant bacteria
Oxepanoprolinamides treat gram-positive and gram-negative bacterial infections in mice
by Bethany Halford
October 28, 2021
| A version of this story appeared in
Volume 99, Issue 40

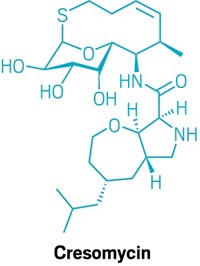
Chemists have developed a new weapon in the ever-escalating arms race against deadly bacteria: a synthetic class of antibiotics called oxepanoprolinamides. Iboxamycin, one of the most promising compounds in the new class, can kill multidrug-resistant bacteria, including methicillin-resistant Staphylococcus aureus and Enterococcus faecalis, in lab tests and can also treat both gram-positive and gram-negative bacterial infections in mice.
Most advances in antibiotics have relied on semisynthesis, in which compounds are made by cells via fermentation and then tweaked chemically to add or modify groups. To make more dramatic changes to an antibiotic’s skeleton, researchers led by Harvard University’s Andrew G. Myers and the University of Illinois Chicago’s Yury S. Polikanov decided to take a fully synthetic approach. They developed chemistry that could couple the aminooctose portion of clindamycin, a semisynthetic antibiotic, to a variety of other groups. The approach allows them to probe how changes in molecular structure affect antibiotic potency (Nature 2021, DOI: 10.1038/s41586-021-04045-6).
The resulting class of molecules, the oxepanoprolinamides, muck up the bacterial ribosome, killing the cells. They do this by binding at the same place on the ribosome that clindamycin does. One way that bacteria develop resistance to clindamycin is by adding two methyl groups to one of the ribosome’s nucleotides, which prevents clindamycin from binding. But Myers and Polikanov’s team found that iboxamycin binds deeply within the ribosome, dislodging the dimethylated nucleotide. Iboxamycin overcomes other types of resistance mechanisms too, but the researchers are still determining precisely how.
Myers says the researchers would have been unlikely to design iboxamycin based on the bacterial ribosome structure information they had when they began this project. The ability to synthesize and test a large array of candidates is a key advantage of this synthetic system. “Antibiotics discovery is still an empirical enterprise,” Myers says. “It’s a numbers game. We make a lot of molecules, and I think that’s an important thing to recognize.”
Lynn Silver, an expert in antibiotic research who runs LL Silver Consulting, says in an email that “further optimization will probably be necessary to improve the potency and antibacterial spectrum of the oxepanoprolinamides, but the class looks very promising.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter