Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Industrial Safety
What did we learn from the Deepwater Horizon disaster?
10 years after the largest accidental oil spill in history, scientists look back on insights gained about how microbes and chemical processes degrade oil. And they still see gaps that need to be filled before the next big spill
by Giuliana Viglione, special to C&EN
September 13, 2020
| A version of this story appeared in
Volume 98, Issue 35

Credit: ©Daniel Beltrá, courtesy of Catherine Edelman Gallery, Chicago | A ship sits on a band of oil from the 2010 Deepwater Horizon spill.
In brief
The Deepwater Horizon blowout caused gas and oil to spill into the Gulf of Mexico for 87 days. Ten years after this environmental disaster, scientists studying the spill better understand the complex processes that affect how oil transforms and degrades in the water and just how resilient ecosystems can be to such a disturbance. The disaster response also did nothing to clear up debates about chemicals called dispersants that are often used in spill cleanups. Researchers hope that the technological advances over the past decade—and the knowledge gained—will better prepare the world for the next big spill.
Early on the morning of April 21, 2010, Samantha “Mandy” Joye awoke to an ominous email. A colleague of hers was out on the Gulf of Mexico on board the R/V Pelican, one of the Louisiana Universities Marine Consortium’s research ships. In the distance, she could see smoke billowing into the sky. There seemed to be more ships on the water than usual, too. With limited internet access on the ship, she asked Joye to figure out what was going on.
That evening, Joye, an oceanographer and microbiologist at the University of Georgia, discovered the cause: an explosion on BP’s Deepwater Horizon oil rig, located about 65 km off the Louisiana coast in an area known as the Macondo Prospect. According to reports that night, 11 workers on the platform were missing; officials later confirmed that the workers had all died in the explosion. As she read the initial descriptions of the explosion, Joye felt her stomach tightening into a knot. The wells in this part of the Gulf are deep below the ocean’s surface. If the undersea safety measures designed to prevent a blowout had failed, it would be difficult to fix any leaks. “Everybody that works out in the Gulf—the fear is always something like that happening,” she says.
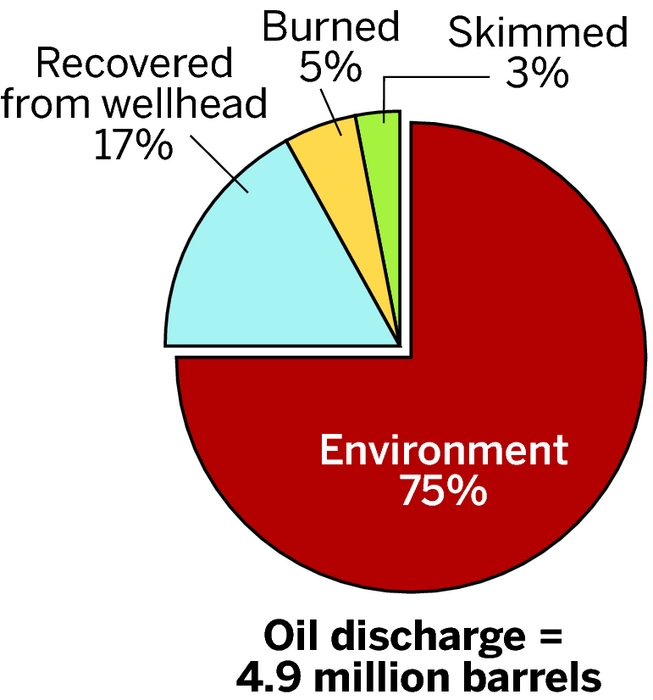
The oil rig sunk the next day. Later that afternoon, the first oil slick, stretching for 8 km along the ocean’s surface, appeared near where the Deepwater Horizon once stood. For the 87 days that followed, the wellhead, damaged by the explosion, spewed petroleum deep below the surface of the Gulf of Mexico. In the end, between 4 million and 5 million barrels of petroleum leaked into the Gulf, making the spill the largest single accidental release of gas and oil into the ocean in history.
Sept. 19 marks 10 years since retired US Coast Guard Admiral Thad Allen, the national incident commander in charge of the Deepwater Horizon response, announced that the spill was over. Although workers had capped the well on July 15, it wasn’t until mid-September that the drilling of the relief well was completed. Cement pumped in through this secondary well permanently sealed the petroleum reservoir.
In the years since the blowout, scientists have studied many aspects of the spill. They’ve tried to measure its ongoing environmental and public health impacts. They’ve attempted to catalog the natural processes by which the oil transformed and degraded. They’ve continued to debate the injection of chemicals called dispersants near the site of the blowout and whether the benefits outweighed the risks. And as funds for researching the disaster have waned, researchers hope to apply the lessons they’ve learned to prepare for the next major spill.
The dispersant debates
The magnitude of the oil spill wasn’t immediately apparent in the days after the rig explosion. It wasn’t until April 24, 4 days after the explosion, that the US Coast Guard announced in a press conference that oil was leaking from the wellhead. Five days after that announcement, Bobby Jindal, then governor of Louisiana, declared a state of emergency. At the beginning of May, says Ed Overton, an environmental analytical chemist and emeritus professor at Louisiana State University, “it got really hectic.”
Most previous spills had been single events—for example, the grounding of an oil tanker and the rapid release of the petroleum on board. The Deepwater Horizon accident had left the wellhead gushing oil constantly, and because the rupture was about 1.6 km underwater, it would be difficult to cap it effectively. Those conditions meant that “you had a new spill every day,” Overton says.
As the oil began to reach the surface, responders turned to a familiar line of defense in oil-spill cleanup: dispersants. These proprietary solutions contain many compounds, including surfactants, emulsifying agents, and solvents. Once sprayed on an oil slick, the dispersants act like a detergent and break the oil into smaller droplets that can then mix into the water, where oil-eating microbes can degrade it. Using dispersants offshore can reduce the amount of oil that washes onto the beaches, protecting both people and fragile coastal ecosystems, Overton says.
But the use of the chemicals has been controversial since they were first deployed in large quantities in the 1967 Torrey Canyon spill off the southwest coast of Great Britain. The dispersants wreaked massive environmental damage, decimating marine organisms including seaweed, plankton, and mollusks. Later analysis revealed that ecosystems in areas treated with the chemicals recovered up to five times as slowly as those that were not. And although the formulations of dispersants have changed significantly in the decades since that spill, many people are still wary of their use. Nalco’s Corexit dispersants, applied to the coastal waters of Alaska during the 1989 Exxon Valdez oil spill, caused many health problems among response workers and disrupted the fisheries and ecosystems that are vital to the Alaskan economy.
A spill's spread

In response to the Gulf spill, engineers suggested injecting dispersants near the underwater wellhead, hoping that the chemicals would break up the spewing oil into smaller, less buoyant droplets that would stay far below the ocean’s surface. There, the oil could eventually be metabolized by microbes, settle to the ocean floor, or get carried out of the Gulf entirely. This approach might also help improve the air quality for response workers on the surface by trapping in the depths some compounds that would have volatilized.
But all existing tests on the efficacy of dispersants had been conducted on the surface of water, says Elizabeth Kujawinski, a chemical oceanographer at Woods Hole Oceanographic Institution. There was no guarantee that they would perform the same way in the crushing depths of the ocean.
Despite these safety and efficacy concerns, the US Environmental Protection Agency authorized the first-ever subsurface use of dispersants on May 15, 2010. Eventually, engineers pumped almost 3 million L of two Corexit formulations below the surface of the Gulf. Aircraft and boats sprayed another 3.8 million L of dispersant to treat the oil on the surface.
Scientists have accounted for the fate of about 80% of the oil released into the Gulf by the blowout. Of that amount, about half made it up to the surface (Proc. Natl. Acad. Sci. U.S.A. 2012, DOI: 10.1073/pnas.1110564109). Many of the light hydrocarbons, which are highly soluble in seawater, dissolved in the water column. And much of the rest of what was released formed deep plumes of oil that eventually stretched more than 32 km from the gushing well—an indication, perhaps, that injecting the dispersants at depth had prevented some oil from reaching the surface.
But the presence of the deep-sea plume did not convince some researchers that the dispersants had done their job. The size and shape of the hole in the well, as well as the large amounts of natural gas in the fluid inside the Macondo reservoir, may just as easily have caused the oil to atomize and form a plume, says Claire Paris-Limouzy, a biological oceanographer at the University of Miami. And when you take into account the toxicity to subsurface organisms like plankton and deep-sea fish, she says, “we should not be using any kind of deep-sea dispersants. That was more catastrophic than not using anything.”
The dispersants also affected the crews that helped clean up the spill. According to surveys of spill-cleanup workers, many who had been exposed to dispersants showed symptoms related to the chemicals, such as skin irritation and respiratory problems, several years after the spill. More than half the exposed workers experienced skin irritation; about a quarter reported a cough or burning eyes or both (Environ. Health Perspect. 2017, DOI: 10.1289/EHP1677). A whistleblower report published by the Government Accountability Project in April 2020 documents ongoing health problems in response workers.
For the general population, dispersants probably didn’t have a big effect, says Xiaoping Pan, an environmental toxicologist at East Carolina University. “But to the ocean organisms, the use of dispersants is devastating.”
In her studies using the model organism Caenorhabditis elegans, Pan has found that both crude oil from the Macondo reservoir and Corexit 9500A inhibit reproductive function, especially when exposure occurs during development. And a mixture of crude oil and dispersant is much more toxic to C. elegans than either of the two components alone (Arch. Toxicol. 2012, DOI: 10.1007/s00204-012-0936-x). Another study that looked at toxicity to a key plankton species found that the dispersant-oil mixtures were more than 50 times as toxic as oil alone (Environ. Pollut. 2012, DOI: 10.1016/j.envpol.2012.09.024).
But other researchers aren’t interested in reigniting debates over the use of dispersants during the disaster. The decision to use the chemicals “was totally reasonable” based on the information available at the time, says Christopher Reddy, an environmental chemist at Woods Hole Oceanographic Institution. “When you’re in a crisis, you’re not afforded the same luxuries as when you’re a bench chemist.”
Where the response went wrong, he says, is in not collecting crucial data, such as air-quality measurements, that could have informed whether the solutions were working as hoped. “It’s unfortunate that we don’t have the data in hand to sway folks one way or the other.”
The evidence in the literature is equivocal as to whether dispersants were effective at depth, Woods Hole’s Kujawinski says. And because the chaotic conditions of the real world are impossible to replicate in a lab, the dispersant debates aren’t going away anytime soon. “Dispersants do remain—and I think will remain—a topic of some controversy until, unfortunately, an experiment can be conducted under appropriate field conditions,” she says. “And none of us want that to happen.”
Comparing spills

Weathering the storm
While the Deepwater Horizon spill continued to feed the debate over dispersants, it also provided scientists with an opportunity to learn more about what happens to oil sitting on the ocean’s surface. Researchers have known that a variety of so-called weathering processes, both natural and human driven, transform and degrade the hydrocarbons in oil over varying timescales on the surface of a body of water. The most volatile components, such as low-molecular-weight alkanes, evaporate off almost immediately. Response crews burn or skim what they can off the ocean’s surface. Oil that is dispersed—either naturally or chemically—is mixed into the water column, where hydrocarbon-degrading microorganisms metabolize it. Some oil sitting on the surface mixes with seawater to form viscous, sticky emulsions. And some fraction of the oil is transformed into partially oxidized compounds by photochemical processes.
Before the Deepwater Horizon spill, scientists didn’t think that oxidation by sunlight played a significant role in oil-weathering processes, Reddy says. “Photochemistry is typically just this little afterthought.” The conventional wisdom was that only those compounds that directly absorb sunlight, such as polycyclic aromatic hydrocarbons (PAHs), could be photooxidized. And these reactions were thought to occur in the weeks or months after a spill, after much of the oil had already been cleaned up by other means.
But when Reddy’s group analyzed oil residues from the spill, they found that about half the surface oil—far beyond the small fraction of crude oil components that are capable of directly absorbing sunlight—had been partially photooxidized. Furthermore, the researchers showed that these reactions likely occurred in the hours and days after the oil reached the surface, with two-thirds of the oxidation occurring within the first 10 days of exposure (Environ. Sci. Technol. 2018, DOI: 10.1021/acs.est.7b05948). Rather than directly photooxidizing, much of the crude oil had been oxidized by the highly reactive oxygen-containing radicals produced when PAHs or other crude oil compounds absorb sunlight—a process called indirect photooxidation. “Once that free radical starts reacting,” Louisiana State’s Overton says, “the first thing it bumps into, it oxidizes.”
These oxidation products mixed with other compounds in the oil-water emulsions, further increasing their viscosity and making them more difficult to clean up from both the ocean’s surface and the coastal marshes, where a lot of them ended up.
Limitations in the equipment used by previous generations of analytical chemists hampered their efforts to understand these products. But advances in high-resolution mass spectrometry and new ionization techniques allowed scientists studying the Deepwater Horizon spill to begin to understand the high-molecular-weight materials that make up these deposits. And as scientists gain the capabilities to further probe these materials, Overton says, “we’re learning a lot more about them.”
Advertisement
The Deepwater blowout also demonstrated the limitations of traditional methods of evaluating the extent of an oil spill, including satellite and aerial imaging or 2-D modeling to predict its spread on the water’s surface.
University of Miami’s Paris-Limouzy and her colleagues developed a new type of model that predicted the path of the oil droplets from the moment they left the wellhead, taking into account their buoyancy, their chemical composition, and the speed at which they would be metabolized. Using that 3-D model, they found that how far PAHs and other toxic compounds spread in the Gulf was likely much larger than the visible extent of the spill as captured by satellite and aerial imagery. Comparing the modeled oil concentrations to previously published data on water and sediment samples showed that the computer’s predicted extent of the oil spread was reasonable.
The new model suggested that toxic oil products reached beyond the area that had been closed to fishers to prevent them from picking up fish contaminated by such compounds. Although the area’s boundaries matched well with the visible oil extent, according to Paris’s model, they captured only about 70% of the area that included toxic compounds (Sci. Adv. 2020, DOI: 10.1126/sciadv.aaw8863).
It wasn’t surprising that the closure area didn’t capture the full extent of the oil’s spread, Paris-Limouzy says. “What was surprising was how much was missed.” In the future, using 3-D models from the onset of a spill could help better inform the response, she says.
A microbial revolution
Below the ocean’s surface, microorganisms degraded the bulk of the oil plumes drifting through the Gulf. And the organisms did so with remarkable speed, says Joel Kostka, a microbial ecologist at the Georgia Institute of Technology. “We’ve been pretty surprised at how fast hydrocarbon degradation can occur as long as there’s plenty of oxygen,” like in the deep waters of the Gulf or the well-aerated sands of the Gulf Coast’s beaches, he says. In other areas, such as seafloor sediments and coastal salt marshes, low levels of oxygen limited microbes’ ability to metabolize oil hydrocarbons. As a result, those areas contain remnants of the blowout. When big storms move through the Gulf and churn up the seafloor, tar balls and other oil residues still wash up on the shores.
Because the Gulf of Mexico has so many natural hydrocarbon seeps, researchers suspected that the microbes endemic to the Gulf might be well adapted to metabolize the leaking fuel—that the body of water was primed for a spill. And indeed, the microorganisms responded rapidly to the injection of natural gas and crude oil. A succession of microbial taxa bloomed and then receded in response to the changing composition of the hydrocarbons from the spill (Environ. Sci. Technol. 2013, DOI: 10.1021/es401676y).
But it’s hard to say for certain whether the Gulf ecosystem is unique in its ability to respond to a massive environmental perturbation like the Deepwater blowout. Hydrocarbon-degrading microbes are “rare but ubiquitous,” having been found at low levels in environments around the world, Kostka says. And while the Gulf of Mexico “seems to have more of an abundance of microbes with this capacity,” he says, the disaster also coincided with a revolution in microbiology via “omics” technologies such as metagenomics and metatranscriptomics. So it’s possible that scientists know of more oil-degrading microorganisms inhabiting the Gulf of Mexico than elsewhere simply because that’s where most of the research has been done.
Metagenomics, the study of the aggregated genetic material in an environment, and the miniaturization of sequencing tools have also led to the rise of forensic environmental microbiology. By searching for hydrocarbon-degrading bacteria, researchers can detect areas affected by a spill after other signals, such as visible oil or its degradation products, have faded away. “Microbiology as a field has a lot to offer response science,” the University of Georgia’s Joye says. “These organisms provide a sentinel of oil that lasts long after the oil may have disappeared.” And because the microbial response to the spill followed a specific pattern, the presence of a particular genus of bacteria can indicate how far along in the response an ecosystem is.
The first microbial samples, taken in late May 2010, showed that certain classes of microbes had grown exponentially in the areas affected by the plume. These microbes were the ones that fed on the relatively light n-alkanes and cycloalkanes, but as that food source declined, so did the bacteria that fed on it. By mid-June, other classes of microbes had begun to bloom in their place. And after the well was capped in mid-July, yet another significant shift in the microbial population occurred as the available food source changed once again.
Although the individual species of microbes populating the Gulf have not returned—and may not return—to their prespill composition, the microbial ecosystem as a whole has essentially been restored to its normal function, Joye says. After the spill, the bolus of natural gas and complex hydrocarbons introduced into the waters took about 5 years to cycle through the Gulf’s microbes and plankton. But other parts of the ecosystem, including deep-sea corals and organisms higher up on the food chain, still show evidence of the blowout’s destruction, such as reproductive failure. “It’s not over yet,” Joye says. “The deepwater environment has not recovered.”
To truly understand how the Gulf ecosystem is recovering, she says, scientists need prespill data to establish a baseline to compare against. And even though the Gulf of Mexico has been heavily researched, scientists never collected a lot of those relevant data before the disaster. So it may be difficult to ever determine whether the Gulf has recovered completely.

What did we learn?
Applying lessons from one oil spill to the next is challenging, Joye says. The necessary cleanup response varies according to any number of factors: the composition of the crude oil, the temperature and microbial ecosystem of the water, the prevailing water currents in the region, and the weather on the days after the spill. Even the Ixtoc I oil spill, the 1979 well blowout in the Gulf of Mexico that provides the closest analog to the Deepwater disaster, didn’t prove that instructive to those cleaning up in 2010. For example, many of the lessons that responders could have learned from the 1979 disaster were recorded in industrial literature rather than in academic journals, making them difficult to access.
Advertisement
Across many fields, experts echo the same refrain: long-term data before a spill are necessary for assessing disaster response. Without baseline data of marine ecosystems, Joye says, it’s nearly impossible to assess the damage to and recovery of those environments. Comprehensive surveys of microbial and fish populations, carried out regularly in areas susceptible to disaster or change, would establish a baseline that could help researchers understand the effects of oil spills or even climate change. And good public health data before and after a disaster would allow other researchers to tease out the potential effects of exposure—to either oil or dispersants—on human health, says Maureen Lichtveld, an environmental health researcher at Tulane University. “Longitudinal cohort studies are the only way we’re going to address some of those lingering questions.”
There are some ways in which Deepwater has prepared the community for the next disaster, Kujawinski says. Technologies that were just beginning to come into their own a decade ago are now commonplace. The advances in mass spectrometry and ionization techniques have rapidly expanded scientists’ understanding of oil-weathering processes. Portable genome sequencers could allow researchers to assess microbial populations in real time to detect the stage of ecosystem recovery. And increasingly complex 3-D ocean models can take chemical, physical, and biological processes into account to predict the extent and the path of the spilled oil.
But these technologies are useful only as long as the people equipped to use them can reach the site of the spill. “When you’re thinking about and planning for future spills,” Reddy says, “how does it work when you don’t have an absolute perfect setup to be in theater and the infrastructure to support it?” In many ways, the Gulf of Mexico was perfectly set up for a spill response. Its currents, its petrochemistry, and its geology were all relatively well studied. And the robust infrastructure of the Gulf states—their vast networks of highways, multiple deepwater ports, more than a dozen airports—meant that it was relatively easy to mobilize response teams and researchers alike.
Not all spills happen in such accessible spots. On July 25, a cargo ship ran into a coral reef off the coast of Mauritius. In the weeks that followed, an estimated 7,333 barrels of oil leaked from the ship into the fragile marine ecosystem. With less spill-response infrastructure on hand, addressing this spill is more difficult than it was in the Gulf. The same will be true for potential future spills in the Arctic as the frozen north is opened up for more oil drilling. So future response technologies should be developed with use by civilians rather than scientists in mind, Reddy says. Because at the end of the day, “this is about people,” he says. “It’s humans that can put the biggest change in the response of a crisis.”
Giuliana Viglione is a freelance writer based in Washington, DC.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter