Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Azacyclic allenes harnessed
Strained system participates in Diels-Alder cycloaddition to construct complex scaffolds
by Bethany Halford
August 5, 2018
| A version of this story appeared in
Volume 96, Issue 32

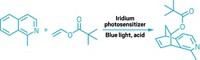
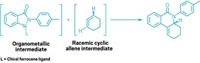
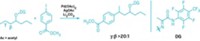
The allene motif—in which two double bonds join three carbons together—tends to be an oddball among functional groups. Squeeze an allene into a ring system, and it becomes even more unusual. A team at the University of California, Los Angeles, led by Neil K. Garg and Kendall N. Houk, hopes to change the allene’s reputation with a report that azacyclic allenes can be useful synthetic tools (Nat. Chem. 2018, DOI: 10.1038/s41557-018-0080-1). The researchers developed silyl triflate precursors that readily generate azacyclic allenes by adding CsF under mild conditions—an advance over past syntheses that required harsh conditions and gave poor yields. These azacyclic allenes undergo Diels-Alder reactions with dienes (example shown). Computational studies of the transition states of these reactions show how substituents are able to control which double bond reacts. The methodology provides access to substituted piperidines—which are often skeletons in drug molecules—by forming two new bonds and three stereocenters with high levels of diastereoselectivity. The chemists also report [3+2] and [2+2] cycloadditions with the azacyclic allenes. “Our present study demonstrates that strained azacyclic allenes, although largely neglected, serve as valuable building blocks for the construction of complex molecular scaffolds bearing multiple stereogenic centers,” they note.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter