Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
C-H Activation
Directing group lets chemists reach remote C–H bonds
New reaction activates γ-C–H bonds over the conventionally favored β-C–H bonds
by Tien Nguyen
April 20, 2019
| A version of this story appeared in
Volume 97, Issue 16

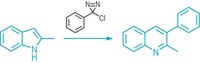
Because of their ubiquity in organic compounds, C–H bonds have long tantalized chemists as synthetic handles. But these bonds are both relatively inert and virtually indistinguishable to reagents, synthetic challenges that have spawned an entire field of research known as C–H activation. One such strategy in this area, known since the 1970s, is to treat a heteroatom-containing molecule with a palladium catalyst, which hooks onto the heteroatom and the neighboring carbon atom to form a thermodynamically favored five-membered palladacycle. But this approach activates only C–H bonds in the β position, the second carbon away from the heteroatom. Now, researchers led by Jin-Quan Yu at Scripps Research have extended the reaction’s reach by one carbon (Nat. Chem. 2019, DOI: 10.1038/s41557-019-0245-6). Because C–H bonds defy the typical tools for controlling selectivity via metal insertion, such as tweaking a catalyst’s electronics or sterics, Yu says, the team turned to ring strain. They proposed that capping alcohol-containing substrates with an imine or pyridone directing group would favor the less-strained 6,5 bicyclic palladacycle over the 5,5 bicyclic intermediates. Using its directing group method, the team preferentially plucked off γ-C–H bonds to install a variety of aryl molecules (example shown). Directing groups were removed by standard hydrogenation or reduction. Now the researchers have their sights set on the seven-membered palladacycle.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter