Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
C-H Activation
Strong, aliphatic C–H bonds succumb to direct arylation
Dual-catalyst approach allows chemists to construct connections between a vast array of aliphatic and aromatic molecules
by Bethany Halford
August 1, 2018
| A version of this story appeared in
Volume 96, Issue 32

While many chemistries have been developed to incite C–H bonds to react, certain types of aliphatics stubbornly resist such transformations. Now, chemists at Princeton University and Merck & Co. have come up with a dual-catalyst reaction that activates aliphatics with recalcitrant C–H bonds, provoking them to react with aryl bromides.
The finding opens new avenues for medicinal chemists, who are always on the lookout for reactions that give them the ability to add complexity to existing drug candidates quickly and easily, says Princeton’s David W. C. MacMillan, who led the research. The reaction will allow medicinal chemists to tweak a compound to improve its properties or to prepare a series of similar compounds to compare how structural modifications alter their activity.
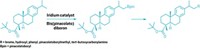
The reaction combines light-driven, polyoxometalate-facilitated hydrogen atom transfer and nickel catalysis (Nature 2018, DOI: 10.1038/s41586-018-0366-x). A decatungstate catalyst generates carbon-centered radicals from strong, neutral C–H bonds. These radicals then act as nucleophiles in nickel-mediated cross-coupling with aryl bromides, forging C(sp3)–C(sp2) bonds between the aliphatic and aryl groups.
“You go into this thinking it’s going to be an unholy mess, but it turns out it’s exquisitely selective,” MacMillan says, adding that the group can predict with 95% accuracy which C–H bond will react. The C–H bond that reacts must yield a stable radical and that radical must be able to attach to the nickel and participate in reductive elimination with the metal.
“The direct cross-coupling of unactivated, aliphatic C–H bonds is one of those transformations that synthetic chemists dream about,” comments Erik Alexanian, an organic chemist who studies aliphatic C–H functionalization at the University of North Carolina, Chapel Hill. The new reaction, he says “is sure to find broad application. It’s a wonderful example of how innovative applications of dual catalysis can unlock solutions to challenging problems in synthesis.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter