Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Medicinal Chemistry
Medicinal chemistry methods miniaturized for high-throughput experimentation
Researchers redesign 4 workhorse reactions to run in microliter droplets
by Mark Peplow, special to C&EN
July 2, 2023
| A version of this story appeared in
Volume 101, Issue 21

For Tim Cernak of the University of Michigan, small certainly is beautiful. He and his colleagues have redesigned some of the most popular reactions in medicinal chemistry so that they work in microliter droplets of solvent, enabling them to be used in the latest robotic high-throughput experimentation (HTE) systems (Nat. Synth. 2023, DOI: 10.1038/s44160-023-00351-1).
HTE allows chemists to run dozens or hundreds of reactions in parallel, each within a tiny well on a plastic plate. In medicinal chemistry labs, HTE can rapidly generate many analogs of a drug candidate for biological testing, and it can also help to optimize reactions by generating vast amounts of data about which combination of conditions offers the best outcomes.
Plates with more wells enable higher throughput, while smaller wells allow chemists to run the reactions on minute quantities of precious starting materials, such as natural products. Some commercial plates boast a staggering 1,536 wells, each with a volume of about 1 µL, and these are used in an extreme version of the technology known as ultraHTE. But miniaturization poses challenges for many reactions—not least because volatile solvents and reagents easily evaporate from the diminutive droplets within these wells. That rules out using common solvents such as tetrahydrofuran or dichloromethane, for example.
To overcome this problem, Cernak and his colleagues modified the conditions of four workhorse medicinal chemistry reactions so that they could run at room temperature in high-boiling- point solvents and were compatible with plastic labware and other equipment used in HTE systems. “We focused on identifying reaction conditions where you could use less-volatile solvents, so that you can miniaturize with ease,” Cernak says.
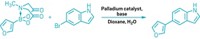
The team used HTE to screen a range of conditions and reagents for the Suzuki-Miyaura coupling, reductive amination, N-alkylation, and N-Boc-deprotection reactions. These four and amide coupling (which the team had miniaturized previously) account for about two-thirds of all the reactions run in medicinal chemistry labs, Cernak says.
Using N-methyl- 2-pyrrolidone as the solvent for reductive amination, for example, the researchers made a library of 46 analogs of staurosporine, an anticancer agent. This required just 100 nmol of the scarce natural product in each reaction.
“The amount of information they get out of small quantities of starting material is amazing,” says Magnus Johansson, a senior principal scientist in medicinal chemistry at AstraZeneca in Gothenburg, Sweden, who uses HTE and was not involved in the research.
For N-Boc-deprotections, Cernak’s team replaced the volatile reagent trifluoroacetic acid with concentrated sulfuric acid and used this method in a two-step sequence to create various analogs of other anticancer compounds. The researchers also showed that all four miniaturized reactions could be scaled up using the same conditions to produce larger amounts of any promising products.
“It’s just a phenomenal resource that reduces the barrier to implementation” of HTE, says Stephen G. Newman at the University of Ottawa, who uses HTE for reaction development and discovery and was not involved in the research.
Much of the work was done while Cernak was in a previous role at Merck & Co., and he says that researchers across the industry are interested in using their miniaturization strategies. Meanwhile, high equipment costs are still a major barrier for academic chemists wanting to access HTE, but Newman hopes the research demonstrates that it could be worth using in their own labs.
“The more cases where you prove that the system is powerful, the more appealing it becomes for others to start entering this area,” Newman says. “This makes it more attractive for people who want to get into this technology, so they can be confident that problems can be solved.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter