Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
ACS Meeting News
Electrons catalyze molecular assembly
Simple catalysts can push molecular recognition and assembly into high gear
by Mitch Jacoby
March 23, 2022
| A version of this story appeared in
Volume 100, Issue 11

A few electrons are all it takes to significantly boost the rate of supramolecular assembly of one molecule threading through another. The finding extends electron catalysis, which is used in synthetic covalent chemistry, to noncovalent chemistry, and may lead to new complex forms of materials.
Non-covalent bonding processes, such as molecular recognition and supramolecular assembly, occur widely in chemistry and biology. The rates of these phenomena, which can be sluggish, can be jumpstarted, but doing so generally requires complex catalytic systems.
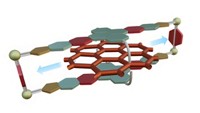
On Wednesday at the American Chemical Society Spring 2022 meeting, Yang Jiao of Northwestern University reported that the assembly rate of a host-guest complex can be increased by a factor of 640 in the presence of simple catalysts—electrons.
Speaking in a session organized by the Division of Organic Chemistry, Jiao described a study involving the assembly of a complex consisting of a ring-shaped molecular host and a dumbbell-shaped guest. The host contains two bipyridinium (BIPY) radical cations. The guest consists of three units; a BIPY radical cation binding site in the center, which drives assembly with the host via radical-pairing interactions; a bulky diisopropylphenyl group on one end that cannot be threaded through the ring; and a dimethylpyridinium (PY) cation on the other end. Under normal conditions, repulsion between the PY cation and the BIPY radical cations prevents the host and guest from assembling.
The researchers found that catalytic quantities of various types of chemical electron sources, including metals, metal complexes, and common reducing agents, rapidly increase the reaction rate with little dependence on the type of source. Applying electric current to a solution of the molecules also catalyzed the assembly process. Adding electrons to the system lowers Coulombic repulsion, enabling the PY end of the dumbbell to thread the ring, as shown by quantum calculations.
Jiao and others, including Northwestern’s Sir J. Fraser Stoddart and William A. Goddard III of the California Institute of Technology, recently published this work in Nature (2022, DOI: 10.1038/s41586-021-04377-3).
Redox-driven formation of mechanically interlocked molecules has been known for a long time, but “the involvement of electron catalysis during the process is remarkable and eye-opening,” said Rafal Klajn of the Weizmann Institute of Science. He hypothesizes that the catalytic activity could be further increased if either the ring or the thread component were surface-immobilized.
CORRECTION:
This story was updated on March 23, 2022, to clarify Rafal Klajn's comment. He thinks the catalytic activity would increase if either the ring or the thread component of the complex were surface-immobilized, not just the thread.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter