Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Treatment For Alcohol Abuse
Experimental drug curbs excessive alcohol use in rats
by Sophie L. Rovner
March 12, 2007
| A version of this story appeared in
Volume 85, Issue 11
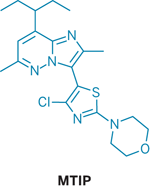
A new compound synthesized by Eli Lilly & Co. showed such promise in animal tests that "it should be developed for clinical use in alcoholism," according to neuroscientist Markus Heilig, clinical director at the National Institute on Alcohol Abuse & Alcoholism (NIAAA). "We're thrilled, because this has been a dream for over a decade for many of us," says Heilig, who led the team that evaluated the compound's effect in rats (J. Neurosci. 2007, 27, 2718).
Heilig found out about the compound, dubbed MTIP, when NIAAA set up a collaboration with Lilly to develop pharmacotherapies for alcoholism. Heilig's team now has tested MTIP in rats that were genetically predisposed to develop alcoholism and in rats that had been trained to be alcohol-dependent. MTIP curbed alcohol abuse in both models, "suggesting that it may be effective in treating a broad range of alcoholics," says Glenn R. Valdez of Grand Valley State University, in Allendale, Mich.
The compound inhibits binding of corticotropin-releasing factor (CRF) to the CRF1 receptor. CRF has long been established as a major regulator of stress in the body and has been linked to various psychiatric conditions, including alcoholism, Valdez explains. "Development of a CRF1 receptor antagonist has been a major goal of many pharmaceutical companies," adds Valdez, who studies the role of CRF in alcoholism. But it's been challenging to create a small-molecule antagonist that can be taken orally and enter the central nervous system, where it can reach the CRF1 receptors. The new drug, which is able to cross into the CNS, "seems to show a lot of promise," he says.
Most previous CRF1 blockers accumulate in the liver, potentially causing toxicity, Heilig says. But MTIP is markedly improved in this regard. "That's a major breakthrough in the chemistry," he notes.
Philip A. Hipskind and his team of Lilly chemists discovered MTIP when they synthesized several compounds designed to block CRF1 activity without being toxic or having other undesirable properties. Donald R. Gehlert and other Lilly colleagues then worked out the compounds' in vitro pharmacology and found that the pyridazine MTIP displayed a promising profile.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter